Colloidal nanoparticles prepared from zein and casein:interactions,characterizations and emerging food applications
Yi Wang,Wusigale,Yangchao Luo
a Department of Nutritional Sciences,University of Connecticut,Storrs 06269,USA
b Key Laboratory of Dairy Biotechnology and Engineering,Ministry of Education;Key Laboratory of Dairy Products Processing,Ministry of Agriculture and Rural Affairs,Inner Mongolia Agricultural University,Hohhot 010018,China
Keywords:Zein Casein Nanoparticles Interactions Applications
ABSTRACT Protein colloidal nanoparticles (NPs) are ubiquitous present in nature and function as building blocks with multiple functions in both food formulations and biological processes.Food scientists are inspired by naturally occurring proteins to induce self-assembly behavior of protein by manipulating environmental parameters,providing opportunities to construct special and expected NPs.Zein and casein,the main proteins derived from corn and milk,are two examples of the most prevalently studied food proteins for nanoarchitectures in recent years.In this article,the compositions,structures,and physicochemical properties of these two proteins and casein derivatives are summarized as well as their interactions and characterizations.Strategies to fabricate zein-sodium caseinate based NPs are critically highlighted and illustrated.Particularly,applications such as encapsulation and delivery of bioactive compounds,producing food packaging for enhanced antioxidative and antimicrobial effects,and stabilization of emulsions to achieve fat replacement.Due to the imperative role of food proteins in diet composition,this review not only provides cutting-edge knowledge for nanoparticle construction but also opens new avenues for eff icient utilization and exploitation of food proteins.
1.Introduction
Nanoparticles (NPs) are defined as small colloidal particles with three dimensions in the nanoscale range.Protein-based NPs are ubiquitously present in nature and are involved in numerous biological functions in association with biocatalysis,formation of structural networks,and molecular recognition [1].They are also one of the essential components of nutritional food materials and an excellent source for food-grade NPs due to their inherent bioactivity,biocompatibility,biodegradability and colloidal properties,including self-assembly behavior,denaturation degree,and hydrophobic and hydrogen inter-and intra-molecular interaction [2].Self-assembled protein NPs ubiquitously existed in various commonly consumed foods,such as casein micelles in milk,soy protein particles in tofu,zein in processed foods made with corn proteins.They not only endow the nutritional and health-beneficial effects of food but also act as the skeleton to maintain the stability of foods [3].Inspired by the naturally occurring colloidal NPs,there has been an outpouring of research interests among food scientists to design and develop functional NPs,which are exploited to deliver nutrients,colors,f lavors and preservatives,as well as to modify the texture,appearance,or stability of foods and food packaging.
Protein solubility is a key factor in assembling protein NPs with desired functional properties.As the most popular prolamin in food industry,zein is commercially extracted from corn endosperm via wet-milling.Its strong hydrophobic property makes it possible to use aqueous alcohol extraction as the most common method for both commercial zein production and zein-based NPs fabrication [4].Novel applications of zein are drawing increasing attention in biomedical and pharmaceutical fields as delivery system than its traditional utilization as coating or film materials.The hydrophobicity of zein NPs can be modulated in combination with polysaccharides(e.g.,chitosan [5-7],dextran sulfate [8],soybean polysaccharide [9]and alginate [10]),and other proteins (e.g.whey protein isolate [11,12],β-lactoglobulin [13],and soy protein isolate [14]),which leads to high physical stabilization and oxidative stability.Proteins have greater diversity in terms of composition,structure and function and their increased popularity enables development of new products [15].Development of protein-protein complex NPs using different proteins also balances the amino acids content and improves nutritional value.As the major component of milk,casein and caseinate (NaCas) usually maintain micellar structure with great emulsifying capacity.Thus,the combination of casein and zein was proposed to better prevent occasional aggregation of zein in aqueous solutions and the products exhibited attractive functions,such as encapsulation and oral delivery,stabilizing and emulsifying,water barrier and packaging.However,to the best of our knowledge,a comprehensive and critical review focusing on the development of NPs based on zein-NaCas interactions has not been published.Thus,the emphasis herein is on structures,fabrication techniques,and characterizations,as well as the emerging research on the applications of zein-NaCas NPs in nutrient delivery,food safety,food packaging,and Pickering emulsions.
2.Composition,structure and physicochemical properties
2.1 Zein
Zein is a class of water-insoluble prolamins from corn,which is generally separated into four components based on the solubility and molecular weight:α-zein (19 and 22 kDa),β-zein (14 kDa),γ-zein (16 and 27 kDa) andδ-zein (10 kDa).All zein components are abundant in hydrophobic and neutral amino acids (e.g.,leucine,proline and alanine,more than 50%),leading to the unique solubility of zein,which is mainly restricted to aqueous solutions of acetone,alcohols,and aqueous alkaline solutions [16].Zein is also regarded as a non-dietary plant protein due to its poor nutritional values without two essential amino acids,lysine and tryptophan.More specifically,α-zein accounts for 70% -85% of total zein mass as a major fraction,which is composed of highly homologous repeating units and thus has a highα-helix content [17].The behavior of zein in aqueous ethanol solutions is almost dependent on the microstructure ofα-zein thanks to the predominantly high content ofα-zein in the commercial zein based on the current extraction methods.For its secondary structure,a repeating unit consisting of tenα-helix is antiparallelly positioned and their ends are connected by glutamine-turns,thus forming an elongated rectangular prism model (Fig.1) [18,19].The top and bottom surfaces of the prism are hydrophilic due to the existence of glutamines,while the other four surfaces remain hydrophobic.These four hydrophobic surfaces are linked by hydrophobic bonds,serving as the major driving force for its extension and further aggregation [20].These accumulations and the rounding ofβ-sheet rings enabled the fabrication of nanospheres or nanotubes [21].
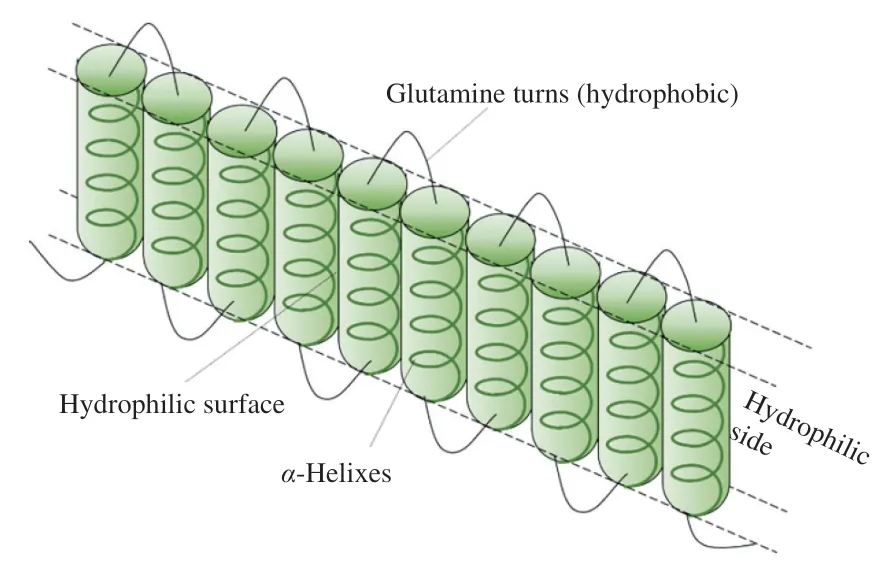
Fig.1 Schematic diagram of zein secondary structure.
While most studies prior to 2010 were primarily focused on the structural properties of zein molecules,there has been a paradigm shift on research into self-assembly behavior of zein and its applications since the past decade.Before self-assembling to NPs at higher concentration,zein firstly forms NPs which is attributed to the opposite polarities of prism shaped structure ofα-zein.Furthermore,the changes of external parameters are contributing to the driving forces of the different microstructures or nanostructures [21].Homogeneous nanoscale zein particles ranging from 100 to 200 nm was created for the first time by manipulating the polarity of zein solution from hydrophobic into hydrophilic rapidly [22].With the evaporation of ethanol,α-helixes were converted toβ-sheets and were packed side by side,forming a long ribbon and eventually curled into rings [16].The coil-shaped structure is supported by the morphology of low concentrationα-zein at neutral pH,showing a globule structure with a height of only 17 nm while the width reaching 120 nm [21].This conformational transition of zein was observed during the evaporation process and measured by circular dichroism (CD) spectra and transmission electron microscopy (TEM) [22,23].Recently,atomic force microscopy (AFM) was applied to characterize its selfassembly behavior and study under AFM had reported the dramatical decrease ofα-helix while the percentage ofβ-sheet and random coil increased during evaporation-induced self-assembly,which is in consistency with the transformation of its secondary structure.These transformation accumulate and theβ-sheet ring round and coil,enabling the fabrication of NPs or nanotubes [21].Interestingly,amyloid fibrils were observed when the zein aqueous ethanol solution was heated,which was not common for prolamins and has not been systematically investigated yet [24].
2.2 Casein
Casein is the major protein in bovine milk,making up 80% of total milk proteins.Four fractions (αs1-,αs2-,β-andκ-casein)are believed to construct the micellar structure,which is a supramolecular structure existing in cow milk [25].Theαs1-casein andβ-casein fractions account for a large proportion of total weight(40% individually),and meanwhileβ-casein is the most hydrophobic fraction and thus is buried in the inner region [26].In contrast,αs2-casein constitutes only 10% of total casein and it contains 11 seryl phosphate groups,which significantly lower the pI from pH 8.3 for non-phosphorylated one to pH 4.9 [27].One of the most unique characteristics ofκ-casein is that nearly half of its molecules in bulk milk are glycosylated to Thr,compared with the other fractions which are generally phosphorylated to Ser [28].Additionally,κ-casein is also the only fraction that has a low calcium sensitivity.Negative charges on casein would decrease due to the bindings of Ca and Mg to Seryl phosphate groups,eventually resulting in instability in aqueous solutions.Whileα-andβ-caseins are recognized as calcium-sensitive caseins [26],casein micelles have great colloidal stability in milk thanks to the steric stabilization effect by the hairy structure ofκ-casein on the outer surface.
Micelles are composed of millions of casein molecules and calcium phosphate,as well as many water molecules for hydration into hydrophilic residues [29].Three models can be abstracted to summarize numerous theories illustrating the structure of casein micelles over the half century.Coat-core model postulated that it is the calcium phosphate nanoclusters that form the core polymers rather than the aggregation ofαs-orβ-casein [30].A porous network of protein was highlighted in the internal structure model with another emphasis on interactions between caseins [29].However,recent experimental data via the ultra-small-angle X-ray scattering method did not support the structural model with interparticle interactions.Instead,such data correlated the casein micelles into a sticky sphere structure [31].Walstra developed the subunit models,depicting a more specific profile of micelles with hydrophobic cores and hydrophilic surfaces [32].Inhomogeneous distribution of casein and water molecules in micelles was stipulated by this model,suggesting the existence of both highly hydrated and poorly hydrated areas.It is proven that casein would break into smaller particles under highpressure homogenization treatment,with a more folded hydrophobic core and increased solubility [33].Though there are controversies over these models,it is widely recognized thatκ-caseins locate on the surface,preventing the aggregation of micelles with their hairy tails.Acidification of cow milk by addition of inorganic acids or lactic acid bacteria can lead to the coagulation of casein micelles and solubilization of calcium phosphate.The production of potassium and sodium caseinate involves three major steps,including removal of whey protein after grinding of acid casein curd,and neutralization via sodium/potassium hydroxide.These ionization processes result in the formation of caseinate salt,thus promoting the solubility of casein in water with a smaller particle size [34].
3.Fabrication strategies,characterizations and interactions of zein-NaCas colloidal NPs
Delivery systems fabricated with polymer-polymer colloidal NPs have received increasing attention in recent years.The polymerpolymer colloidal NPs-based delivery systems combine the advantages of multiple polymers,such as slower release,delayed digestion,and enhanced mucoadhesive property.The NPs fabricated through different methods vary considerably in their physicochemical and structural properties,which determines their gastrointestinal fate and stability.Consequently,different mechanisms and interactions (driving forces) are involved in the formation of NPs,including electrostatic interactions,hydrophobic interactions,hydrogen bonds,and van der Waals force,etc.The main principle to fabricate zein-NaCas NPs is to manipulate their solubility and miscibility by altering their dispersion medium,including solvent concentration,pH condition,temperature,ionic strength,etc.Various fabrication techniques that have been studied so far in literature to prepare zein-NaCas NPs are summarized in Table 1,along with the detailed introduction of each technique in the following section (Fig.2).
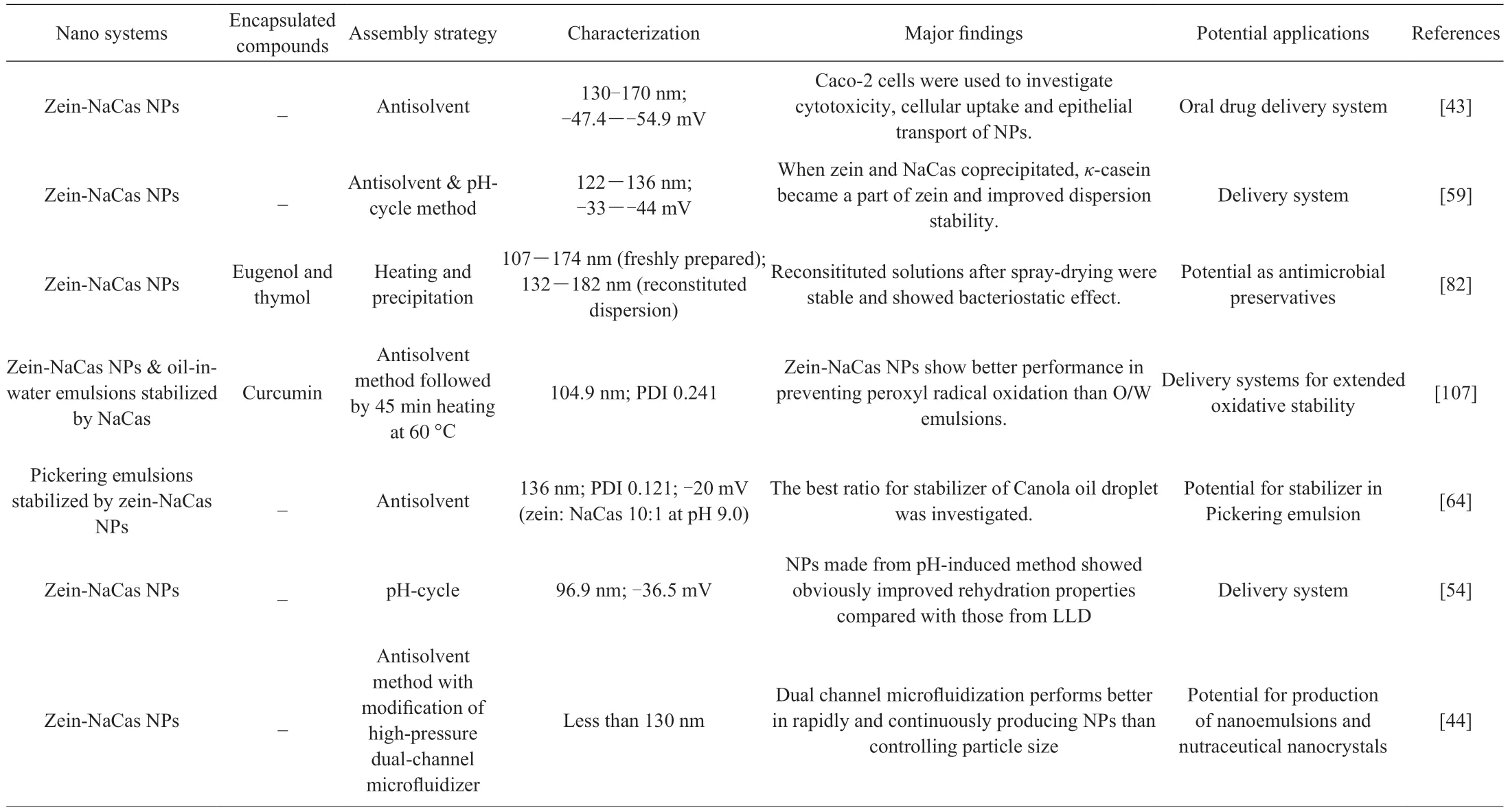
Table 1 Summary of the studies on the delivery systems based on zein-NaCas NPs.
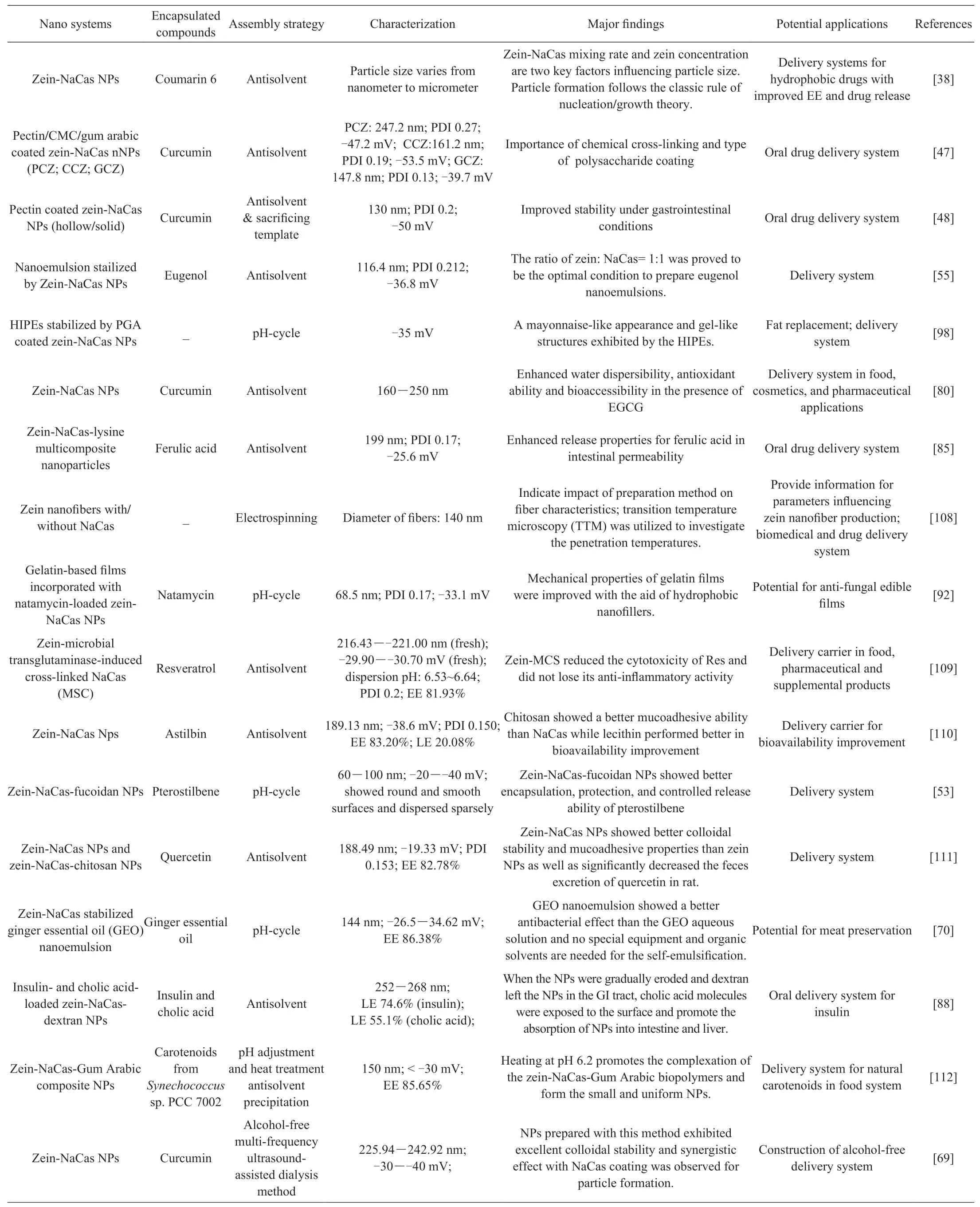
Table 1 (Continued)
3.1 Fabrication strategies
3.1.1 Antisolvent method
A simple,rapid,and cost-effective technique to produce protein colloidal NPs with tunable properties is antisolvent or liquid–liquid dispersion method.This method is based on the injection of a protein solvent mixture into an antisolvent,thereby leading to protein selfassembly and particle formation [35].The particle stability can be improved by adsorbing other biopolymers or surfactants onto their surface to increase electrostatic and steric repulsion,or reduce hydrophobic attraction [36].For zein,ethanol is commonly used as the solvent phase,being the best solvent to produce small particles with uniform sizes than isopropanol and methanol,and water is used as the antisolvent phase [37].Casein has been explored as an efficient stabilizer to form zein-NaCas NPs.NaCas was usually dissolved in the antisolvent phase prior to NPs production (Fig.2A) or added after zein NPs fabrication (Fig.2B),followed by rotary evaporation to remove ethanol [38].This method has been extensively used for the delivery of hydrophobic bioactive compounds through premixing them with zein solution.By co-dissolving in the aqueous ethanol solution at lipophilic bioactive compound to zein ratios of 0.1 (m/m),the bioactive compounds co-precipitate with zein during antisolvent precipitation process,which enables the spontaneous encapsulation and self-assembly of NPs [39-41].In general,the zein-NaCas NPs with well-defined size range (100-300 nm) could be obtained at zein concentrations of 0.25% -2%,and at casein to zein ratios ≥ 1:1 [38,39,42,43].Zein and casein exhibited an isoelectric point at pH 6.2 and pH 4.6,respectively,whereas the isoelectric point of zein-NaCas NPs was observed to be around pH 5 [42].In this case,a pH away from the isoelectric point (pH 5) should be considered to prepare the NPs with great colloidal stability.NPs fabricated via this method have an ideal re-dispersibility after freeze-drying,with no significant change of particle size [40].Ebert modified the fabrication process by using one-step dual-channel microfluidization [44].In this study,zein in ethanol and casein in water was separately pumped and passed through two channels at high velocity,followed by homogenization and thus achieving continuous production of zein-NaCas NPs.The formation of these particles is a result of the shear,turbulence,and cavitation force generated by the high pressure,facilitating the large-scale commercial manufacture.To further improve the stability of zein-NaCas NPs,bio-safe crosslinkers such as microbial transglutaminase and oxidized dextran were applied to create covalent linkage with two proteins [45,46].NPs formed by proteins tend to show poor colloidal stability performance after long time storage and less resistance for gastrointestinal enzymes,leading to a quick release of encapsulated compounds.Due to these drawbacks,polysaccharides,e.g.pectin,gum Arabic,or xanthan gum,were introduced to play a protective role for the core substrates and were either mixed with NaCas solution before particle formation or added after formation of zein-NaCas NPs (Figs.2A and B) [46-50].
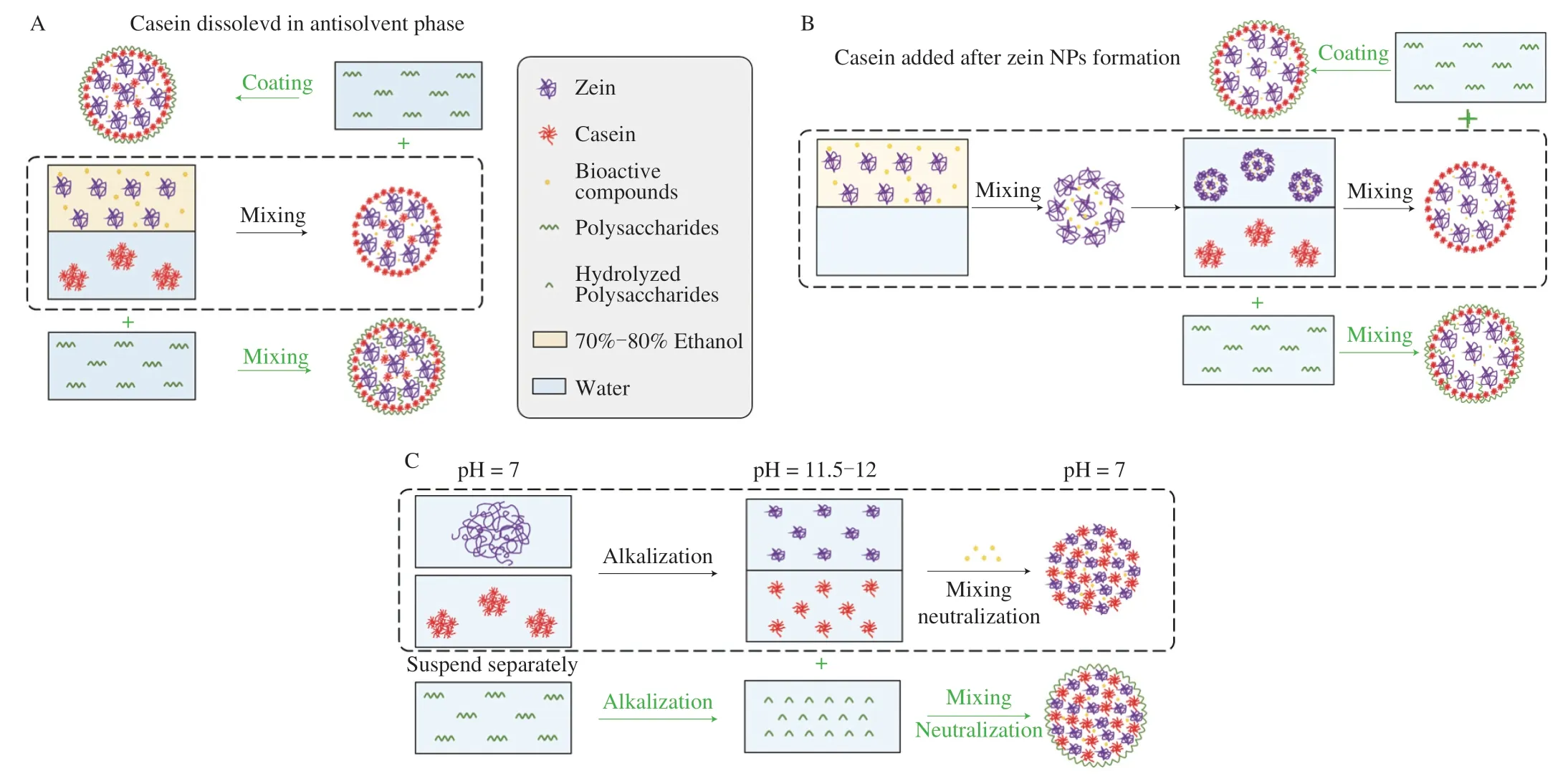
Fig.2 Illustrations of formation mechanisms and possible structures of zein-NaCas and zein-NaCas/polysaccharides NPs fabricated through (A) traditional antisolvent,(B) antisolvent and (C) pH-cycle methods.
3.1.2 pH-Cycle method
To reduce the utilization of alcohol and circumvent the potential inflammatory hazard,pH-cycle method was recently developed as an alternative to the antisolvent method (Fig.2C).It is also recognized as pH-driven or pH-induced approach,which is based on the modulation of pH from alkaline to neutral in control of the self-assembly behavior of proteins.Deprotonation of amino acid residues (e.g.glutamate in zein,lysine and tyrosine in casein) at alkaline condition results in net negative charges and hence causes the dissolution of zein and dissociation of casein micelles [51,52].Thanks to the pH-dependent solubility of zein as well as the self-assembly property of casein,both zein and casein are readily dispersed in alkaline solutions,while the following neutralization facilitates the formation of zein-NaCas NPs.Compared with antisolvent method,zein-NaCas NPs prepared with this approach has a smaller particle size,which was usually less than 100 nm [43,53].Since bioactive compounds like curcumin are insoluble at neutral pH but can be dissolved at alkaline pH due to deprotonation,they could be co-dissolved with zein and caseins and encapsulated in self-assembled NPs during acidification.Although proteins are dissociated to a higher extent at a higher pH,which may provide more sites for protein-protein interaction,the hydrolysis of proteins under strong alkaline condition should be considered [51,52].Typically the pH of protein solutions is initially brought to 11.5-12.0.After mixing the zein and casein solutions and incubation for 30 min,the mixtures are acidified to pH 7 [54].It has been reported that a NaCas to zein mass ratio of 1:1 was sufficient to produce stable colloidal particles [55].Bioactive compounds (e.g.eugenol,curcumin) have been added into this system at loading ratios ranging from 0.2% -2.0% [55,56].The structural changes of the proteins as well as the encapsulated bioactive compounds after the alkaline treatment were negligible [54,57],and thus the pH-induced method at this mild treatment conditions could be used to produce protein-based colloidal particles.However,the method is timeconsuming since it needs titration from high alkaline pH to neutral pH.Moveover,this technique is limited to encapsulated bioactive compound that can be solubilized and maintained bioavailability at alkaline condition.To meet with this problem,recently,partially alkaline hydrolysis of propylene glycol alginate has been used to reduce pH from 12.5 to 7.5,providing a promising alternative to prepare zein-NaCas NPs without the process of titration [58].
3.1.3 Other methods
Researchers have attempted to form zein-NaCas-based colloidal particles using other methods,such as pH-and heat-induced,ultrasound-assisted dialysis method,polydopamine (PDA)-mediated,and structural inversion methods.Zein-NaCas NPs prepared by antisolvent and pH-cycle methods have great re-dispersibility after freeze-drying,while the NPs exhibit poor re-dispersibility after spray-drying.A previous study showed that spray-dried zein-NaCas NPs produced by incorporating the two conditions,i.e.,hot aqueous ethanol and alkaline pH could re-disperse in water with good stability during storage [59].In this method,zein and NaCas were co-dissolved in hot 50% aqueous ethanol adjusted to pH 8.0 before antisolvent precipitation to form NPs.By virtue of the universal adhesive properties,PDA has been widely employed as a coating material for the construction of flexible and multifunctional nanoparticle architectures through non-covalent interactions or covalent interactions.The preparation of PDA-mediated zein-NaCas NPs was based on a modified method of antisolvent precipitation.Briefly,dopamine hydrochloride was dissolved into Tris-HCl buffer acting as antisolvent solution to disperse zein ethanol solutions followed by the addition of casein.Since PDA was easily formed due to the spontaneous oxidation of dopamine,after coating on zein NPs,the functional groups of PDA could conjugate with that of casein to form more stable and robust NPs which were collected by centrifugation and redispersed with deionized water [60,61].Coating by zein could also be achieved by structural inversion method.At lower than 90% ethanol,hydrophobic moieties of zein molecules clump together to form micelle-like structures,while the orientation of each molecule will be reversed when the solvent medium turns more hydrophobic [62].The hydrophilic part of zein aggregates interacts with NaCas,and adsorption and orientation of zein on surface of NaCas film may provide the film with compact structure and improved barrier properties [63].
3.2 Characterizations and interactions
After the preparation of NPs,the evaluation for their quality is based on characterization results,including particle size,surface charge,morphology,structure,and stability test.Results from dynamic light scattering (DLS) not only provide the average particle size of NPs but also the particle size distribution.The typical diameters for zein-NaCas NPs fall within 150-240 nm under the near neutral conditions [22,46,64,65].Smallest particle size was usually observed at the zein: NaCas mass ratio of 1:1 or 1:2,while the higher or lower ratios led to an insufficient or excessive NaCas,being unable to the compact monolayer coating on the surface of zein NPs.Scanning electron microscopy (SEM),TEM,and AFM are three common tools to acquire morphological information of zein-NaCas NPs.Sizes measured under electron microscope are generally smaller than that tested from DLS due to the hydration effect in solutions [17].The shape and surface roughness of zein-NaCas particles are usually indicated from SEM with an observation of homogeneous spherical particles compared with collapsed and red-blood cell shape structures of the pure zein NPs loaded with essential oils [66].AFM characterizes the surface morphology and the three-dimensional structure of NPs since it has an advantage in depicting self-assembly process at nanometer range.By comparison,TEM reveals core-shell structure through a dark-bright color contrast,suggesting desirable adsorption of casein to the surface of zein particles [21,54].Zeta potential,a measurement for surface charge of colloidal suspensions,can be monitored using a zetasizer,presenting electrostatic repulsion and aggregation tendency among colloidal particles.Zein-NaCas NPs prepared by antisolvent or pH-cycle method usually had a zeta potential ranging from -35 mV to -40 mV and showed strong colloid stability [44,59].With the increased amount of NaCas,the absolute value of zeta potentials of these NPs firstly decreased and then kept constant.Zein-NaCas mass ratio of 1:2 was regarded as the optimal condition for the completely surrounded zein in NaCas coating [53].Additionally,since the compounds entrapped into the NPs are usually susceptible to environmental stresses,e.g.,temperature variation and exposure to oxygen,the stability assays are increasingly taken into consideration for a comprehensive evaluation.Compared with the conventional stability test including a long time storage under certain temperature,centrifugal stability test accelerates the whole process by centrifugation and detection of the precipitates [59].The incorporation ability of NPs into food matrix and the shelf life of the encapsulation system can be reflected by the re-dispersibility study.Most researchers reported a desirable storage stability and re-dispersibility after freeze-or spray-dry procedure [67,68].
Intermolecular interactions between the two proteins as well as between the carriers and encapsulated compounds are usually performed by Fourier transform infrared spectroscopy (FTIR),fluorescence spectroscopy,and X-ray diffraction (XRD).The shifts of the characteristic bands in amide I (1 700-1 600 cm-1,mainly C=O stretching) and amide II (1 600-1 500 cm-1,C–N stretching coupled with N–H bending) are associated with the transformation of the secondary structure of zein as well as the formation of hydrogen bonds.The bands at 3 000-2 800 cm-1provide the information on hydrophobic interactions [9].Hydrogen bonds and electrostatic interactions are generally recognized as driving force for the formation of hybrid NPs [53,69,70].Fluorescence spectroscopy can be used to test the surface hydrophobicity of NPs using a hydrophobic probe (8-anilino-1-naphthalene sulfonate (ANS)),and the change in microenvironment of molecules after encapsulation can also be elaborated [47,59].XRD has been employed to investigate the crystal structures of NPs and the encapsulated bioactive compounds.The decrease in peak intensity of polymers after particle formation and that of bioactive compounds after encapsulation suggest the formation of the amorphous complexes [71].In addition,other techniques have been used to characterize NPs,including1H nuclear magnetic resonance,isothermal titration calorimetry,size exclusion chromatography,gel electrophoresis,and liquid chromatographymass spectrometry [6,59,72,73].
Each of these techniques listed above provide information on molecular interactions based on their specialty.Combining these pieces of information might therefore obtain an in-depth mechanism of particle formation as well as interaction among the components.Commonly,the formation of zein-NaCas NPs includes two steps.First,zein molecules self-assemble into NPs accompanied by the encapsulation of the bioactive compounds mainly driven by hydrophobic interactions and hydrogen bonding.Second,zein molecules or NPs interact with casein molecules through hydrophobic,electrostatic and steric repulsion to form the complex NPs or core–shell NPs.In the case of casein addition after zein NPs fabrication by antisolvent precipitation method,adsorption of casein on the zein particle surface,rather than direct complexation is the most likely mechanism [42].However,when casein is dissolved in the antisolvent phase prior to particle production,the mechanisms of direct complexation and co-assembly of zein and casein cannot be ignored [38].The mechanism of forming zein-NaCas NPs during the pH cycle is that casein molecularly interacts and co-assembles with zein molecules through hydrophobic interactions,and the hydrophilic portions of casein molecules provide both steric and electrostatic repulsions to NPs [54].
4.Applications
4.1 Encapsulation and delivery system
Developing colloidal delivery systems for bioactive compounds has become a major research area [74].Since the practical application of bioactive compounds with known health-promoting effects are often hampered by their limited stability and poor water solubility,it is worth exploring zein-NaCas NPs to improve their bioaccessibility and bioavailability.Table 1 summarizes the zein-NaCas NPs that have been reported as delivery systems for different bioactive compounds.
4.1.1 Polyphenols
Polyphenols extracted from plants are increasingly recognized as good for human health and nutritional well-being due to their abundance in phenolic hydroxyl groups and strong antioxidant ability.Curcumin is one of the polyphenols well-known for its antioxidant,anticancer,and anti-inflammatory ability as well as protective effects for even nerval and hepatic cells [75].Its low aqueous solubility and poor bioavailability after oral administration,however,pose a limitation for its absorption and thus hamper its application in functional food.Since 2010,zein was introduced to encapsulate curcumin with the stabilizer of NaCas,and recently,curcumin has been developed as a model compound for studies on encapsulation and release profile of protein-based nanocarriers due to its wide range of sources and obvious orange-yellow color.Patel et al.[76]firstly used curcumin as a probe bioactive compound to investigate interactions between curcumin and zein-NaCas nano-carrier and cellular evaluation,though similar research has been done to assess the cellular uptake and transport of zein NPs coated with NaCas in the absence of bioactives [43].Its solubility increased significantly from 2.88% (free curcumin) to 60% (entrapped in NPs),explained by the involvement of hydrophobic and hydrogen bindings formed between curcumin and zein during the particle formation [77].Zein-NaCas NPs also significantly improved the stability of curcumin under 1 h ultraviolet (UV) irradiation and thermal treatment,with data indicating that the remaining curcumin increased to 93.41% and 66.05% under respective treatment.Moreover,mucoadhension study showed a high retention rate (more than 60%) in 150 min,which may be beneficial to enhance the absorption of curcumin by maintaining the NPs in the proximity around cell membrane for extended period of time,especially the upper GI tract which is the main absorption area [76].
In recent years,protein–polysaccharide colloidal NPs have gained increasing attention for their protective impact on protein cores under gastrointestinal conditions.Sodium alginate,has been utilized to fabricate zein-NaCas/alginate NPs through pH-cycle and antisolvent methods [58,77].The ability to scavenge ABTS free radicals was improved by sixfold for curcumin encapsulated in the NPs when its concentration was greater than 10 μg/mL.Both solid and hollow zein-NaCas/pectin NPs exhibited a more stable kinetic release profile of curcumin and retained 40% curcumin after incubation in simulated gastric fluids for 2 h and simulated intestinal fluids for 4 h [48].The antioxidant ability of curcumin in carboxymethyl cellulose (CMC)coated NPs was nearly fivefold higher than free curcumin dissolved in ethanol [47].Dextran,oxidized by sodium periodate,was then proposed as not only coating material but macromolecular crosslinker to form Schiff base complex with protein cores via covalent linkages,which serve critical interactions to prevent disassembly under stressed condition (such as drying) and thus improve colloidal stability and redispersion.Interestingly,a smaller particles size was also observed with the addition of high concentration (190 nm) of oxidized dextran than that of the low concentration (255 nm) [46].Green tea polyphenol,epigallocatechin gallate (EGCG),is well-known for its function in preventing cancer and cardiovascular diseases [78,79].It is recently reported that the combination of EGCG and curcumin has a synergistic effect on anticancer activity and EGCG would attach between zein and casein layers,thus serving as both a bioactive compound and a crosslinker [80].
4.1.2 Essential oils
Essential oils extracted from natural plants are gaining increasing attention in the replacement of synthetic food additives for their antioxidant and antibacterial abilities.However,given their poor water solubility,most essential oils have limited applications in aqueous systems,where most of food products are based on.While many proteins are amphiphilic biopolymers,zein and NaCas are particularly more hydrophobic and best suited for encapsulation of essential oils.For instance,thymol,the most abundant compounds in thyme,was incorporated into zein-NaCas NPs with a mean particle diameter and zeta potential of 206.0 nm and -44.2 mV at a thymol-tozein ratio of 0.1 (m/m).All these particles had a polydispersity index(PDI) less than 0.16,suggesting a homogeneous size distribution of the dispersion.A slow-release of thymol from such NPs reaching 44% accumulatively after 6.5 h dissolution in deionized water was observed though AFM analysis presented the existence clusters of NPs [81].Eugenol,another essential oil abundant in cloves,has been encapsulated into zein-NaCas-pectin NPs.In this study,dry powder of this nanocomplexes with a size distribution of 1-1.5 μm was obtained from nano spray-drying.The spray-dried powder was able to achieve the same concentration as their original status and similar spherical morphology and smooth surface [65].In addition,Chen investigated the combination effect of encapsulated thymol and eugenol and properties of reconstituted dispersion after spraydrying.A size increase by 10-30 nm and a larger size distribution of rehydrated samples was detected compared with fresh dispersions.This change could be mainly explained by the slight evaporation of essential oils and reconstruction during spray-drying.FTIR result also provided information to prove that interactions between essential oils and protein coatings were totally physical instead of chemical.Encapsulation efficiency at pH 8.0 after spray-drying reached 21.49% and declined from pH 8.0 to pH 6.0,caused by the larger particle size at lower pH.The rapid formation of a semi-permeable protein layer around separated droplets could be achieved easily by smaller particles at pH 8.0 to avoid the evaporation loss of thymol and eugenol [82].
4.1.3 Active pharmaceutical ingredients
One of the challenges limiting the pharmaceutical industry is to improve the dispersion and adhesion ability of lipophilic active ingredients in digestion tract and further enhance theirin vivoperformance.Various encapsulation systems have been designed to tackle this challenge [83].Antisolvent is one of the most common methods for their preparation,which enables the facile encapsulation of hydrophobic compounds,such as dihydromyricetin and ferulic acid,into zein-NaCas NPs [84,85].For instance,Li and colleagues prepared fucoxanthin (FX)-loaded zein-NaCas composite NPs via this method and investigated the anti-proliferative activity of FX,a carotenoid from marine algae which has been proved to have antitumor activity from the induction of apoptosis [86,87].HL-60 cancer cells were used for the evaluation of their inhibitory effect,suggesting the cell viability of encapsulated FX was nearly 20% lower than that of free FX at a concentration of 1 μg/mL [87].In another study by Ahmed,zein-NaCas NPs were applied to encapsulate another anti-hyperlipidemic agent,simvastatin,aiming to lower its dose and minimize its side effects by improving oral bioavailability.Pharmacokinetic studies in rats showed that the maximum plasma concentration (Cmax) for simvastatin zein-NaCas NPs was 23.57 μg/mL,which was 9.05 μg/mL higher than that of free agent.In this study,NaCas functioned as a coating layer to provide a larger adhesion surface between zein particles and the adsorption site in digestion tract [41].Recently,hybrid zein NPs stabilized by casein are designed to overcome the oral absorption obstacle of insulin drug in gastrointestinal tract [88].Exenatide (EXE),a kind of insulinotropic peptide for type 2 diabetes treatment,cannot get access to the intestinal villi and so failed to be absorbed by the intestine epithelium [89].Bao and co-workers [90]successfully constructed an EXE-loaded NPs.This system contained enhancer (cholic acid),protector (zein,Hypromellose phthalate,and phospholipid),and stabilizer (casein).The NPs protected the loaded EXE from degradation in simulated gastric fluid and promoted the intestinal absorption of EXE via bile acid transporters.
4.2 Food safety
Natural antimicrobials have been authorized as food additives and extensively utilized to extend the shelf life of various foods.The double-layer protein structure not only provides more sites for effective binding with preservatives but also prevents them from degradation under extreme environmental stresses [91].Similarly,in another study on natamycin,the zone of inhibition enlarged significantly upon increasing their concentration againstAspergillusniger,Botrytiscinerea,andPenicilliumcitrinum(Fig.3) though no obvious improvement in antibacterial capacity was observed compared with free natamycin [92].Moreover,the growth curve ofE.coliwas also determined to evaluate antimicrobial activity of two encapsulated essential oils of thymol and carvacrol,thus presenting an obvious reduction of 0.8-1.8 lg (CFU/mL) [93].This coencapsulation can not only deliver a sufficient amount of essential oils to the surface of pathogens but also adjust the flavor profile from the individual essential oils.Furthermore,the ginger essential oil-loaded zein-NaCas NPs were prepared into nanoemulsions that were able to reach and disperse in the cell membranes ofPseudomonas aeruginosauniformly,resulting in the damage of cell surface and thus shriveling and wrinkling of the cells.These nanoemulsions also exhibited the improved preservation ability on chilled chicken breasts,a proteinrich model food,preventing the microbial growth and thus meat spoilage [70].

Fig.3 Antifungal activities of the control (CK) and the nanocomposite films GNP3 (0.6 mg/mL of total protein and 0.06 mg/mL of natamycin),GNP5 (1.4 mg/mL of total protein and 0.14 mg/mL of natamycin),and GNATA (film with addition of 0.14 mg/mL of natamycin suspension without NPs encapsulation) against(A) Aspergillus niger;(B) Botrytis cinerea,and (C) Penicillium citrinum;The lesion diameters of different samples against three molds (D).This figure has been reprinted from Ref.[92]with permission from Elsevier.
4.3 Pickering emulsions
Emulsion stabilized by solid particles is defined as Pickering emulsion [94].Though it was originally proposed in the early 20thcentury,it has not been a research focus until last decade.By comparison with traditional emulsions,Pickering emulsion exhibits superior stability during long time storage and is free from the underlying toxicity of synthetic surfactants especially when widely used in food industry.Stabilizers,such as proteins and polysaccharides,are able to adsorb onto the interfacial surface by reducing interfacial energy and provide steric hindrance against droplet coalescence and Ostwald ripening [64,95].Likewise,the colloidal NPs made from protein and polysaccharide exhibit similar surface properties and may act as the colloidal stabilizers to prepare Pickering emulsions.To do so,zein-NaCas NPs are firstly fabricated to constitute an aqueous phase,to which oils are added upon homogenization,enabling the adsorption of colloidal zein-NaCas NPs onto the interfacial layer acting as the stabilizer.In 2012,de Folter and coworkers [96]initially demonstrated the first use of zein in stabilizing oil-in-water (O/W) emulsions,with a droplet size between 10-200 μm.However,this system showed no resistance to creaming in merely three days [96].In another similar study by Gao [95],sodium stearate was applied as a low molecular weight surfactant to enhance the wettability of zein particles,thus obtaining the better performance of emulsions in accelerated stability test [97].Moreover,to avoid the safety concern exposed by small-sized emulsifiers,Feng et al.[64]replaced sodium stearate with NaCas through antisolvent technique.The interfacial properties were characterized visually by confocal laser scanning microscope (CLSM).Upon increasing zein to NaCas ratio from 10:0 to 10:3,the interfacial adsorption of zein-NaCas NPs was markedly improved,resulting in a densely packed monolayer surface coverage and larger droplet size with better stability.Interestingly,as the ratio reached 10:4,NaCas started to adsorb on not only zein as expected but also oil droplet,indicating an excessive amount of NaCas (Fig.2) [64].Compared with the 40% -50% oil fraction as in traditional Pickering emulsion,the research group of Zhong [98]prepared high internal phase emulsions(HIPEs) with the aid of propylene glycol alginate.In this study,phase separation was not observed as the oil fraction rose from 40% to 80% .Nevertheless,when the oil fraction reached 81%,the HIPEs emulsion converted from O/W to water-in-oil (W/O).A higher oil volume fraction resulted in a remarkably stronger rheological modulus and the emulsion tended to show solid-like properties,which was in agreement with previous studies (Fig.4).As CLSM results showed,individual oil droplet was immersed into continuous aqueous phase and packed closely,thus proving the formation of Pickering emulsion [98].Being able to increase the functionality and free from synthetic emulsifiers,Pickering emulsion has achieved a wide application in various industries.For example,this system has been reported to serve as a curcumin carrier to improve itsinvivoantioxidant or work as ingredients to form food packaging films.Recently,increasing attention has been focused on seeking substitutes for partially hydrogenated oils (PHO),which contribute to most texture and mechanical properties in food products.Liu et al.[100]have reported the HIPEs prepared with wheat gluten,displaying similar properties as mayonnaise.While such potential applications of zein-NaCas NPs are to be further validated,current literatures provide proof-ofconcept ideas that zein-NaCas NPs-stabilized Pickering emulsions and HIPEs exhibit excellent colloidal properties for developing fat replacer.Such innovations hold promising potential to revolutionize manufacturing technology for healthy food products with surfactantfree and reduced-fat formulations.
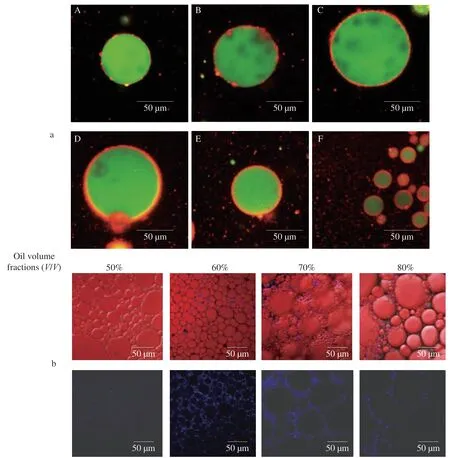
Fig.4 CLSM pictures of emulsions: (a) prepared with plain zein NPs (A),zein-NaCas NPs at zein:NaCas ratio of 10:1 (B),10:2 (C),10:3 (D),10:4 (E),and plain NaCas NPs (F) at pH 3.0 [64](reprinted with permission from Elsevier);(b) stabilized by zein-NaCas/propylene glycol alginate (PGA) NPs at oil volume fractions of 50%,60%,70%,and 80% after storage at 21 °C for 1 day [106](reprinted with permission from American Chemical Society).
4.4 Food packaging
Edible films from food grade sources are promising alternatives for plastic packaging thanks to their nontoxicity,eco-friendliness and barrier properties [101].Packaging fabricated from food proteins,such as zein and NaCas,has been investigated for their enhanced antimicrobial and antioxidant ability as discussed above [56,81].Filmforming solutions containing zein-NaCas NPs and glycerol are often casted onto the glass plates coated with a polyethylene layer to form the packaging film [102].In the study of Yin et al.[63],two different film-forming methods were compared,i.e.,structural inversion and direct coating.Zein dispersion was poured onto the pre-prepared NaCas film and the ethanol concentration was elevated to over 90% to induce self-assembly of zein,while direct formation did not involve the change of solvent polarity.As the concentration of ethanol increased to over 90%,the micelle structure of zein aggregates was observed with 60 s ultrasonic treatment [62].Oxygen permeability test presented an improvement in oxygen barrier properties by structural inversion and this film could be even comparable to the well-known oxygen barrier plastics,e.g.,Ethylene Vinyl Alcohol (EVOH),which may be explained by the hydrophobic interactions of zein coatings.A gelatin-based film incorporated with zein-NaCas NPs was recently proposed.SEM results showed no individual aggregate particles were observed which may be a result from their small size and thereby excellent colloidal stability (Fig.5).Three-phase contact angle (θ),indicating the surface hydrophobicity,is another important criterion for the evaluation of packaging materials.The formation of hydrogen bonding involved of terminal carboxyl and amino was accounted for the decrease of surface hydrophobicity and improvement in thickness and tensile strength [92,103].There are several emerging methods to prepare packaging film for foods,such as electrospraying,airglow discharge plasma and microfluidics [102,104,105],which however are yet to be studied for the preparation of zein-NaCas NPs-based film.In the future,innovative testing methods,such as puncture test,should also be taken into consideration for a more comprehensive evaluation of zein-NaCas incorporated packaging from a food industry perspective [101].

Fig.5 The surface (A) and cross-section (B) of the gelatin film incorporated with the Natamycin-Zein-NaCas NPs observed by SEM [92](reprinted with permission from Elsevier).
5.Conclusions
The unique secondary structures and hydrophobic properties offer zein opportunities for being the potential vehicles to deliver lipophilic bioactive compounds.Casein,being the major component in dairy product,has attracted great attention in exploring its unstructured micelle forms as well as self-assembly and dissociation behaviors within various food systems.Intermolecular bonds and covalent conjugations are the main driving forces of the effective assembly of zein and casein,thus forming NPs featured with a hydrophobic core and hydrophilic coating.One of the major applications of these zein-NaCas based colloids is to effectively deliver lipophilic compounds via oral administration or function as nanocarriers to achieve antimicrobial effects.Zein-NaCas stabilized emulsions,casein-coated zein films and fibers are novel nanostructures that have shown exceptional long-term stability and antioxidant capacity,albeit their mechanical properties may need to be further improved with additional fillers.
Although great emphases have been placed on the preparation and characterization of zein-NaCas NPs,it is still a challenge for food scientists to depict a clear picture of the digestive profile of these particles in gastrointestinal tract.A deep understanding on the microscopic structure of zein-NaCas mixture will also undoubtedly provide fundamental knowledge for the optimization of fabrication strategy.In particular,little is known about the effect of polysaccharide coating on the size and morphology of zein-NaCas NPs and how this shell interacts with inner proteins.Furthermore,future successes in food particulate structures will require knowledge of their rheological information for both microstructure analysis and scale-up production.There are no sufficient reports demonstrating thein vivotoxicity test after being consumed orally,leading to a knowledge gap for animal studies to verify its long-term safety.Last but not least,the possible changes of texture and sensory features in food matrix after incorporation with NPs should also be evaluated and modulated,if necessary,to meet consumer’s preference and expectations.
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
- 食品科学与人类健康(英文)的其它文章
- Biological factors controlling starch digestibility in human digestive system
- Preparation methods,biological activities,and potential applications of marine algae oligosaccharides: a review
- Development of hyaluronic acid-based edible film for alleviating dry mouth
- Mushroom β-glucan and polyphenol formulations as natural immunity boosters and balancers: nature of the application
- Preparation of multicore millimeter-sized spherical alginate capsules to specifically and sustainedly release fish oil
- Structural characteristics,anticoagulant and antithrombotic mechanism of a novel polysaccharide from Rosa Chinensis Flos

