Association of polymorphism in cell death pathway gene FASLG with human male infertility
Deepika Jaiswal, Sameer Trivedi, Neeraj K Agrawal, Kiran Singh*
1Department of Molecular and Human Genetics, Banaras Hindu University, Varanasi, India
2Department of Urology, Institute of Medical Sciences, Banaras Hindu University, Varanasi, India
3Department of Endocrinology and Metabolism, Institute of Medical Sciences, Banaras Hindu University, Varanasi, India
Association of polymorphism in cell death pathway gene FASLG with human male infertility
Deepika Jaiswal1, Sameer Trivedi2, Neeraj K Agrawal3, Kiran Singh1*
1Department of Molecular and Human Genetics, Banaras Hindu University, Varanasi, India
2Department of Urology, Institute of Medical Sciences, Banaras Hindu University, Varanasi, India
3Department of Endocrinology and Metabolism, Institute of Medical Sciences, Banaras Hindu University, Varanasi, India
ARTICLE INFO
Article history:
Received 10 July 2014
Received in revised form 15 February 2015
Accepted 12 March 2014
Available online 20 June 2015
Infertility
Sperm
Apoptosis
Spermatogenesis
Objective:To investigate -844C>T single nucleotide polymorphism (SNP) present in the promoter of cell death pathway gene FASLG with male infertile phenotype.Methods:Genotyping for SNP FASLG (rs763110) was done by polymerase chain reaction followed by analysis with specific endonuclease (PCR-RFLP). DNA sequencing was used to ascertain PCRRFLP results.Results:FASLG -844C>T polymorphism, allele and genotype distribution did not differ significantly between patients and controls (OR: 1.03, 95% CI= 0.7638 to 1.3952, P=0.83). Thus SNP-844C>T of the FASLG gene is not associated with male infertility risk in the analyzed patients.
Conclusion:Human male infertility is a complex disorder and thus other genetic or environmental factors may be contributing to the complex etiology, and further study in other region of Indian populations will verify whether it is associated with male infertility risk.
1. Introduction
Infertility affects 10%-12% couples worldwide and male factor accounts for nearly half of the infertility cases[1, 2]. The formation of mature spermatozoa is a complex phenomenon and requires homeostasis of many different types of cells within the testis[3]. It is regulated by number of genes involved in different pathways like cell proliferation, differentiation and apoptosis[4]. The process of spermatogenesis involves the renewal and differentiation of spermatogonial stem cells into rapidly proliferating spermatogonia, meiotic cells (spermatocytes) and haploid cells (round, elongating and elongated spermatids) before release of a spermatozoon[5]. But to maintain the number of cells in the seminiferous tubules a dynamic balance between cell proliferation and apoptotic cell death is pre-requisite[6]. Apoptosis is involved in a range of physiological functions, such as regulating cell numbers and eliminating unwanted or potentially dangerous cells during the development of organisms, as well as in some pathological processes. Programmed germ cells death is a frequent event occurring during normal spermatogenesis[7]. The extrinsic pathway of apoptosis is key regulatory mechanism for apoptosis in testis. Fas and FasL express on germ cell and Sertoli cells respectively[8]. Expression of FasL is up-regulated in the testes of patients with Sertoli cell only (SCO) and maturation arrest (MA), which suggests that it may be associated with apoptotic elimination or altered maturation of Fas-expressing germ cells through the activation of caspase-3[9]. Fas/FasL expression in the human testis is developmentally regulated and it may be involved in quality control mechanism of the sperms[10]. The FASLG -844 C>T (rs763110) functional polymorphisms is located in thebinding motif of transcription factors disrupt CAAT/enhancerbinding protein. Higher basal expression of FASLG is statistically significantly associated more with the FASLG -844C allele than with the FASLG -844T allele[11]. Based on the biological and pathologic consequence of FASLG, it is possible that functional genetic variations in the FASLG gene may contribute to the clinical outcomes of male infertility. Therefore, we evaluated the effect of the FASLG-844 C>T functional variant of FASLG in cases of idiopathic male infertility.
2. Material and methods
2.1. Subjects
Patients were recruited from the Out Patient Department of University hospital, Infertility Clinic, Institute of Medical Sciences, Banaras Hindu University, Varanasi, India. The present case-control study consists of 204 idiopathic azoospermic infertile males (32.0 ± 4.8 years) and 217 fertile men of comparable age were taken as controls. Both cases and controls belong to same geographical location and have same ethnicity. Patients married for a minimum of two years, having unprotected intercourse were considered for the present study. Three semen analysis were carried out after three/ four days of sexual abstinence to ascertain their infertility status. The size, volume and consistency of testis, occurrence of varicocele, hydrocele or absence of secondary sexual characters were also recorded. Questionnaire was maintained for each patient to record details of their lifestyle, habits and family history. Informed consent was obtained from every participant of each group. Patients with obstructive azoospermia, hypogonadism, hypoandrogenism, chronic diseases, history of pelvic/spinal injuries, karyotype abnormalities and AZF microdeletions were excluded. The control group consists of healthy fertile males who have at least one child and no history of chronic illness. Approval of the University’s ethical committee for research on Human material was obtained.
2.2. FASLG-844 C>T polymorphism genotyping
FASLG-844 C>T polymorphism was studied by polymerase chain reaction (PCR) restriction fragment length polymorphism (RFLP). The C>T change at -844 position creates a restriction site for BsRD1 restriction endonuclease. Two previously described primer sequences were used for PCR: For (5’- CAGCTACTCGGAGGCCAAG -3’) and Rev (5’- GCTCTGAGGGGAGAGACCAT -3’)[11]. The PCR conditions were 30 seconds at 94 ℃, 55 seconds at 62 ℃, and 55 seconds at 72 ℃. The first cycle was preceded by 5 minute denaturation step and a single step of extension at 72 ℃ for 10 minutes followed the last cycle. The 401 bp PCR product was digested with 1 Unit of BsRD1 at 55 ℃ over night. Separation of the products was performed on a 2% agarose gel.
2.3. Sequencing
PCR product was sequenced with Taq-Dye deoxy-terminatorcycle sequencing kit (Applied Biosystem, USA) using an automated genetic analyzer 3130 sequencer to ascertain PCR-RFLP results. Reverse primer was used for FASLG -844 for cycle sequencing. The Sequence was aligned and analyzed using AB DNA Sequencing Analysis software (version 5.2).
2.4. Statistical analysis
Allele and genotype distribution among groups were evaluated using Chi-square test. The difference in frequency between the case and control groups was analyzed for statistical significance at the 95% confidence interval using χ2test. The allele frequency FASLG -844 C>T was in Hardy-Weinberg equilibrium. Odds ratios (ORs) were calculated and reported within the 95% confidence limits. All statistical tests were two-sided, and P-value < 0.05 was considered statistically significant.
3. Results
A total of 421 individuals (204 infertile patients and 217 fertile controls) were examined. PCR-RFLP analysis was performed to assess the genotypes of 844 C>T polymorphism in the promoter region of FASLG gene in cases and controls (Figure 1). The genotype and allele frequencies for 844 C>T polymorphism in the FASLG gene were compared between the two groups. For FASLG -844, T allele frequency in infertile patients was 35.3% and in controls was 29.5%. In the current study, there was no substantial difference in the distribution of both C and T allele between patients and controls (OR: 1.03, 95% CI= 0.7638 to 1.3952, P=0.83) (Table 1). However dominant model (TT vs. CT+CC) showed significant increased risk with male infertility.
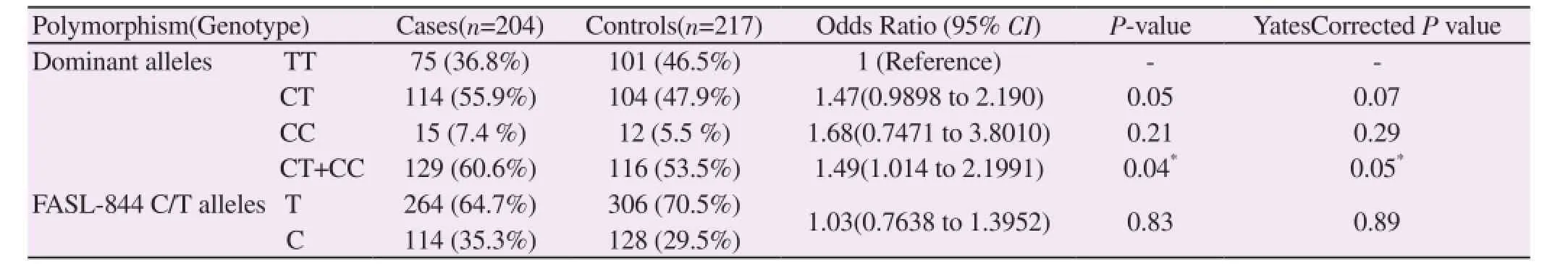
Table 1 Distribution of genotypes and allele frequencies of C844T SNP of FASLG in the study population.
4. Discussion
Spermatogenesis is a dynamic and synchronized process of maturation of spermatogonia into mature spermatozoa. The spermatogonia cells proliferate to give more number of cells, then differentiate into spermatocytes and finally mature sperm[12]. In the testis, Fas-induced apoptosis plays an important role during spermatogenesis. Fas (APO-1, CD95), a transmembrane receptor protein, transmit an apoptotic signal within cells when bound by Fas ligand[12]. Fas and FasL express on germ cells and Sertoli cells respectively. Studies have shown, when the testicular environment can no longer support spermatogenesis, the FasL system is activated leading to germ cell apoptosis, a process in which the Sertoli cells play a major role[7]. Testicular germ cells which are defective or in excessive number externalize Fas, when they come in contact of FasL on sertoli cells, undergo apoptosis and are resorbed. Thus, apoptosis plays an important role in (i) selecting germ cells and (ii) maintaining a proper amount of germ cells available for maturation by the Sertoli cells. Quality of semen is related with fertilization rate, implantation and development of offspring[13, 14]. Studies using the generalized lymphoproliferative disease (gld) mice, which express a nonfunctional form of FasL further illustrate the importance of the Fas signaling pathway in the regulation of spontaneous germ cell apoptosis as well as increased germ cell death after toxicantinduced Sertoli cell injury[15,16]. Apoptosis is an essential process involved in range of physiological functions and some pathological processes. In the present study, functional polymorphism in cell death pathway gene FASLG was genotyped to investigate its role in human male infertility. In addition to this study Wang et al[17] have shown that FASLG-844C/T SNP may be a genetic predisposing factor of idiopathic azoospermia or severe oligozoospermia among Han Chinese men. In contrast, Guixiang et al[18] did not find any association of FASLG 844 C>T with altered sperm apoptosis and poor semen quality in Chinese population. The mutant C allele was found in a statistically identical prevalence among controls and patients. In the present study we have not found any association of FASLG 844>T SNP with male infertile phenotype in North Indian population. In the present study we have found association of FASLG -844 T SNP with male infertile phenotype in North Indian population using dominant genetic model (TT vs. CT+CC). In addition to this study Wang et al have shown that FASLG-844C/T SNP may be a genetic predisposing factor of idiopathic azoospermia or severe oligozoospermia among Han Chinese men. In contrast, Ji et al did not find any association of FASLG 844 C/T with altered sperm apoptosis and poor semen quality in Chinese population. The mutant C allele was found in a statistically identical prevalence among controls and patients. Human male infertility is a multifactorial trait thus both genetic as well as environmental factors contribute to its etiopathology. In summary, our study underscores the significance of genetic variant in the regulatory regions of FASLG in modulating risk of male infertility.
Conflict of interest statement
We declare that we have no conflict of interest.
Acknowledgments
We are thankful to all the patients for providing blood samples. Financial assistance in form of Senior Research Fellowship by Council of Scientific & Industrial Research (CSIR), New Delhi, India to the first author is acknowledged.
[1] O’Flynn O’Brien KL, Varghese AC and Agarwal A. The genetic causes of male factor infertility: a review. Fertil Steril 2010; 93: 1-12.
[2] Makker K, Agarwal A and Sharma R. Oxidative stress & male infertility. Indian J Med Res 2009; 129: 357-67.
[3] Huckins C. The morphology and kinetics of spermatogonial degeneration in normal adult rats: an analysis using a simplified classification of the germinal epithelium. Anat Rec 1978; 190: 905-926.
[4] Lee J, Richburg JH, Younkin SC, Boekelheide K. The Fas system is a key regulator of germ cell apoptosis in the testis. Endocr 1997; 138: 5.
[5] Singh K, Jaiswal D. Human male infertility: A complex multifactorial phenotype. Reprod Sci 2011; 18: 418-25.
[6] Suda T, Takahashi T, Golstein P, Nagata S. Molecular cloning and expression of the Fas ligand, a novel member of the tumor necrosis factor family. Cell 2003; 75: 1169-1178.
[7] Lee J, Richburg JH, Shipp EB, Meistrich ML, Boekelheide K. The Fas system, a regulator of testicular germ cell apoptosis, is differentially up regulated in Sertoli cell versus germ cell injury of the testis. Endocr 1999; 140: 852-858.
[8] Rodriguez I, Ody C, Araki K, Garcia I, Vassalli P. An early and massive wave of germinal cell apoptosis is required for the Development of functional spermatogenesis. EMBO J 1997; 16: 2262-2270.
[9] Kim SK, Lee HJ, Yang H, Kim HS, Yoon YD. Prepubertal exposure to 4-tert-octylphenol induces apoptosis of testicular germ cells in adult rat. Arch Androl 2004; 50: 427-441.
[10] Wu J, Metz C, Xu X, Abe R, Gibson AW, Edberg JC, et al. A novel polymorphic CAAT/enhancer-binding protein β element in the FasL gene promoter alters Fas ligand expression: a candidate background gene in African American systemic lupus erythematosus patients. J Immunol 2003; 170: 132-138.
[11] Russell LD, Warren J, Debeljuk L, Richardson LL, Mahar PL, Waymire K G, et al. Spermatogenesis in Bcl-w deficient mice. Biol Reprod 2001; 65: 318-332.
[12] Brinkworth MH. Paternal transmission of genetic damage: findings in animals and humans. Int J Androl 2000; 23: 123-135.
[13] Jensen TK, Carlsen E, Jorgensen N, Berthelsen JG, Keiding N, Christensen K, et al. Poor semen quality may contribute to recent decline in fertility rates. Hum Reprod 2002; 17: 1437-40.
[14] Nagata S, Golstein P. The Fas death factor. Science 1995; 267: 1449-1455.
[15] Richberg JH, Nanez A, WilliamsLR, Embree ME, Boekelheide K. Sensitivity of testicular germ cells to toxicant-induced apoptosis in gld mice that express a nonfunctional form of Fas-ligand. Endocr 2000; 141: 787-793.
[16] Wang W, Lu N, Xia Y, Gu A, Wu B, Liang J, et al. FAS and FASLG polymorphisms and susceptibility to idiopathic azoospermia or severe oligozoospermia. Reprod Biomed 2009; 18: 141-147.
[17] Ji G, Gu A, Hu F, Liang J, Xia Y, Lu C, et al. Polymorphisms in cell death pathway genes are associated with altered sperm apoptosis and poor semen quality. Hum Reprod 2009; 24: 2439-2346.
ment heading
doi:10.1016/S2305-0500(15)30006-3
*Corresponding author: Kiran Singh, Assistant Professor, Department of Molecular & Human Genetics, Banaras Hindu University, Varanasi-221005, India.
Tel: +91-542-6702489, 09454210058
Fax: +91-542-670-2499
E-mail: singhk4@rediffmail.com, skiran @bhu.ac.in
 Asian Pacific Journal of Reproduction2015年2期
Asian Pacific Journal of Reproduction2015年2期
- Asian Pacific Journal of Reproduction的其它文章
- Potential of pre-gestational intake of Laportea interrupta L. (stinging nettle) leaf decoction as an aid for fetal-maternal health
- Antifertility potential of hydroalcoholic extract of Cordia dichotoma G Forst. leaves: A folklore medicine used by Meena community in Rajasthan state in India
- Effect of genistein on proinflammatory cytokines and estrogen receptorβ in mice model of endometriosis
- Effect of Piper betel leaf stalk extract on protein metabolism in reproductive tissues of male albino rats
- Hepatoprotective mechanism of lycorine against carbon tetrachloride induced toxicity in swiss albino mice - A proteomic approach
- Antitumor activity of aqueous extract of Ziziphus jujube fruit in breast cancer: An in vitro and in vivo study
