The effect of intravitreal ranibizumab after pars plana vitrectomy in severe proliferative diabetic retinopathy
Bo Chen, Xian Zhang, Hua-Jing Yang, Zhi-Qi Chen, Shuai-Shuai Wang, Tie-Mei Yin, Xu-Fang Sun
Department of Ophthalmology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, Hubei Province, China
·Original article·
The effect of intravitreal ranibizumab after pars plana vitrectomy in severe proliferative diabetic retinopathy
Bo Chen, Xian Zhang, Hua-Jing Yang, Zhi-Qi Chen, Shuai-Shuai Wang, Tie-Mei Yin, Xu-Fang Sun
Department of Ophthalmology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, Hubei Province, China
Received: 2016-04-18 Accepted:2016-09-09
•AIM: To observe the results of intravitreal injection ranibizumab after pars plana vitrectomy in severe proliferative diabetic retinopathy.
•METHODS: In this retrospective non-comparative interventional case series, twelve patients had undergone pars plana vitrectomy with silicone oil tamponade, perfluoropropane tamponade or balanced salt solution tamponade. At the end of the surgery, ranibizumab was injected into vitreous cavity.
•RESULTS: Average follows up was 2.75mo. Twelve eyes include vitreous haemorrhage (1/12), vitreous haemorrhage with fibrovascular proliferation (1/12), tractional retinal detachment with vitreous haemorrhage (3/12), tractional retinal detachment with fibrovascular proliferation (2/12), tractional retinal detachment with vitreous haemorrhage and neovascular glaucoma (1/12), tractional retinal detachment with vitreous haemorrhage and fibrovascular proliferation (2/12), tractional retinal detachment with vitreous haemorrhage and fibrovascular proliferation and neovascular glaucoma (1/12), traction-rhegmatogenous retinal detachment with vitreous haemorrhage (1/12). Among these 12 eyes, 8/12 had silicone oil tamponade, 2/12 had perfluoropropane tamponade, and 2/12 had balanced salt solution tamponade. All the patients did not have any therapy before. The retinal attachment rate was 10/10. One patient(1/12)had hyphema after surgery. Postoperative best-corrected visual acuity improved in 9 eyes (9/12), was unchanged in one (2/12), decreased in (1/12) eyes. Eight eyes (8/12) did not have diabetic macular edema after surgery from the optical coherence tomography examination.
•CONCLUSION: Intravitreal ranibizumab after pars plana vitrectomy is useful in severe proliferative diabetic retinopathy. It can improve the success rate of surgery; improve postoperative best-corrected visual acuity; minimize the frequence of diabetic macular edema; minimize the frequence of operations and postoperative complications.
diabetic retinopathy; vitrectomy; ranibizumab
INTRODUCTION
Diabetic retinopathy (DR) especially the severe proliferative diabetic retinopathy (PDR) is a leading cause of vision loss in population worldwide[1]. Despite the success of pars plana vitrectomy (PPV) in managing the severe PDR, significant postoperative complications still occur leading to surgery failure and blindness. Ranibizumab is a recombinant humanised antibody that inhibits human vascular endothelial growth factor (VEGF)[2]. It has been administered intravitreally in VEGF-mediated disease processes such as choroidal neovascularization, age related macular degeneration (AMD), PDR and so on. This study is a retrospective series of cases treated with ranibizumab that was injected at the end of the PPV for the management of severe PDR.
SUBJECTS AND METHODS
We retrospectively reviewed the data for 12 patients. These 12 eyes underwent 23G PPV and silicone oil (SO), perfluoropropane (C3F8) or balanced salt solution (BSS) tamponade at Tongji Hospital, Wuhan, Hubei Province, China between Jun. 2014 and Feb. 2015. At the end of the surgery, ranibizumab (0.05 ml/0.5 mg) (Lucentis, Genentech Inc., USA) has been injected into vitreous cavity (Figure 1). All patients underwent complete ophthalmological examination including visual acuity testing [early treatment of diabetic retinopathy study (ETDRS), best corrected visual acuity (BCVA)], slit lamp biomicroscopy, intraocular pressure (IOP) measurement, and indirect ophthalmoscopy before and after surgery. If patient’s fundus could be visualized, optical coherence tomography (OCT) and fundus photography would be done.
RESULTS
Twelve eyes (12 patients): 5 males (41.7%) and 7 females (58.3%); Surgical indications were: vitreous haemorrhage (VH) (1/12, 8.3%),VH with fibrovascular proliferation (FVP) (1/12, 8.3%) (Figure 2), tractional retinal detachment (TRD) with VH (3/12, 25.0%), TRD with FVP (2/12, 16.7%) (Figure 3), TRD with VH and neovascular glaucoma (NVG) (1/12, 8.3%), TRD with VH and FVP (2/12, 16.7%) (Figure 4), TRD with VH and FVP and NVG (1/12, 8.3%), traction-rhegmatogenous retinal detachment (TRRD) with VH (1/12, 8.3%). The length of follow up was mean 2.75mo (Table 1).
Preoperatively, all the patients were phakic.Two of twelve (16.7%) phakic eyes underwent lensectomy at the time of surgery (all were left aphakic). Cyclocryotherapy was done intraoperatively in 1 (8.3%) eye. One (8.3%) eye has been done the cyclophotocoagulation after surgery. Membrane peeling was done in all eyes. Ten of twelve (83.3%) eyes have retinal detachment (RD), so SO (8 eyes) or C3F8 (2 eyes) were filled into vitreous cavity. SO was removed in 1 (8.3%) eye at 3mo.
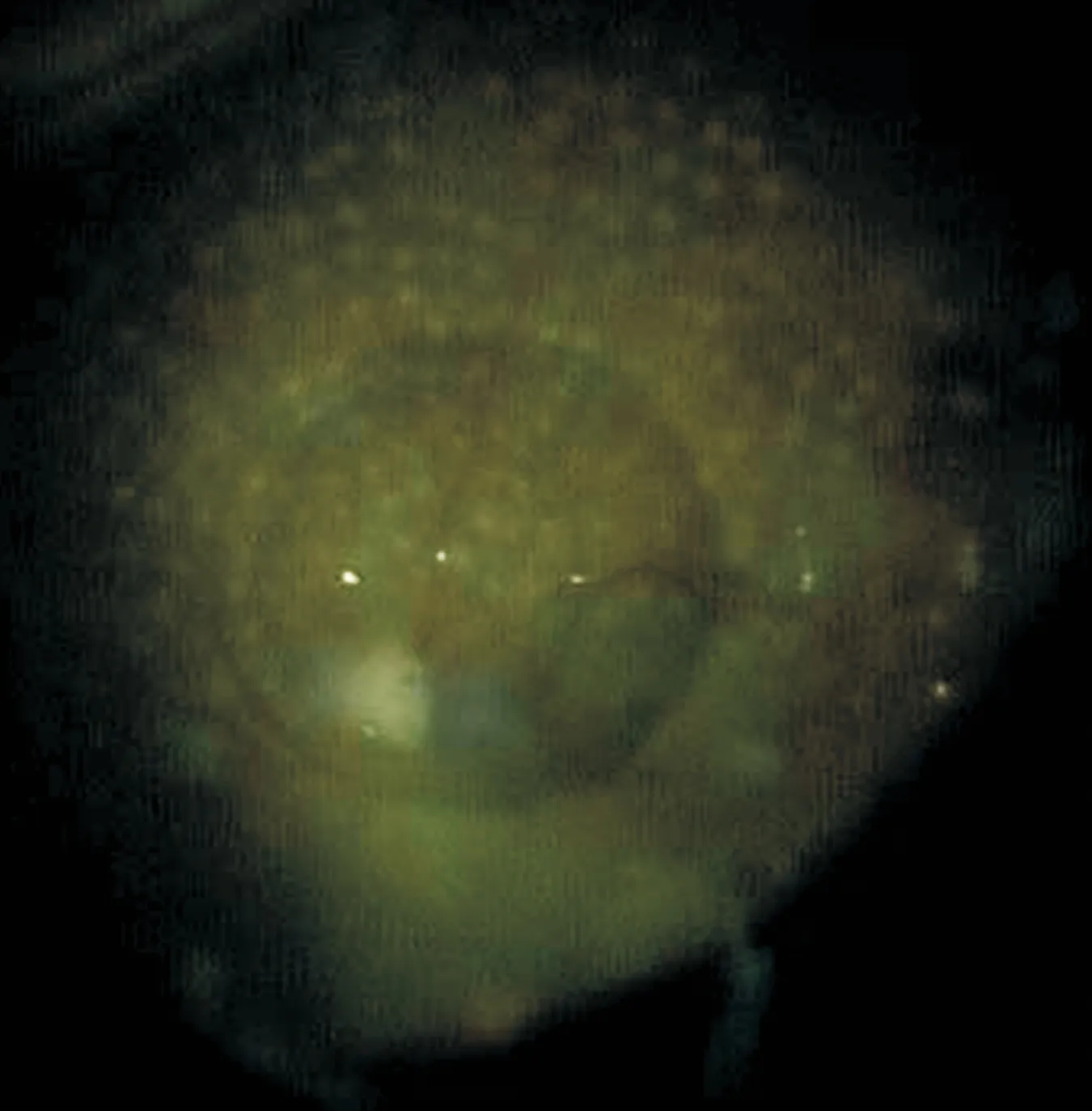
Figure 1 Fundus photograph demonstrates a ranibizumab droplet in a silicone oil-filled vitreous cavity, taken immediately after ranibizumab injected into oil-filled vitreous cavity.
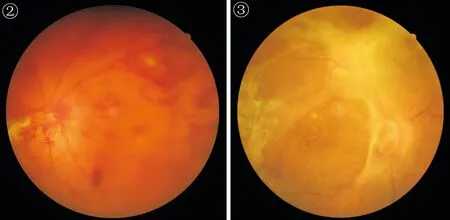
Figure 2 Color fundus photograph demonstrates patient with vitreous haemorrhage with fibrovascular proliferation.
Figure 3 Color fundus photograph demonstrates patient with tractional retinal detachment with fibrovascular proliferation (the same patient with Figure 1).
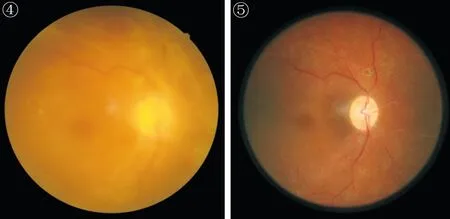
Figure 4 Color fundus photograph demonstrates patient with tractional retinal detachment with vitreous haemorrhage and fibrovascular proliferation.
Figure 5 Postoperative color fundus photograph shows the macula and the posterior pole devoid of any neovascularization. Intraoperative endolaser panretinal photocoagulation is evident (the same patient with Figure 4).
Anatomical attachment was achieved in 10/10 (100%) of the entire RD eyes (Figure 5).
Postoperative BCVA improved in 9 eyes (75%), was unchanged in 2 (16.7%) and decreased in 1 eye (8.3%). Both eyes had postoperative vision of light perception. The decreased eye has severediabetic macular edema (DME) after surgery. Eight of twelve (66.7%) eyes did not have DME after surgery for 1mo from the OCT examination (Figure 6).Postoperative complications included cataract (1 eye), and hyphema (1 eye). Rubeosis iridis (RI) was present preoperatively in 2 eyes, all of them haveneovascular glaucoma (NVG).
Table 1 Patient’s basic information, functional and anatomical status after PPV and intravitreal ranibizumab for PDR
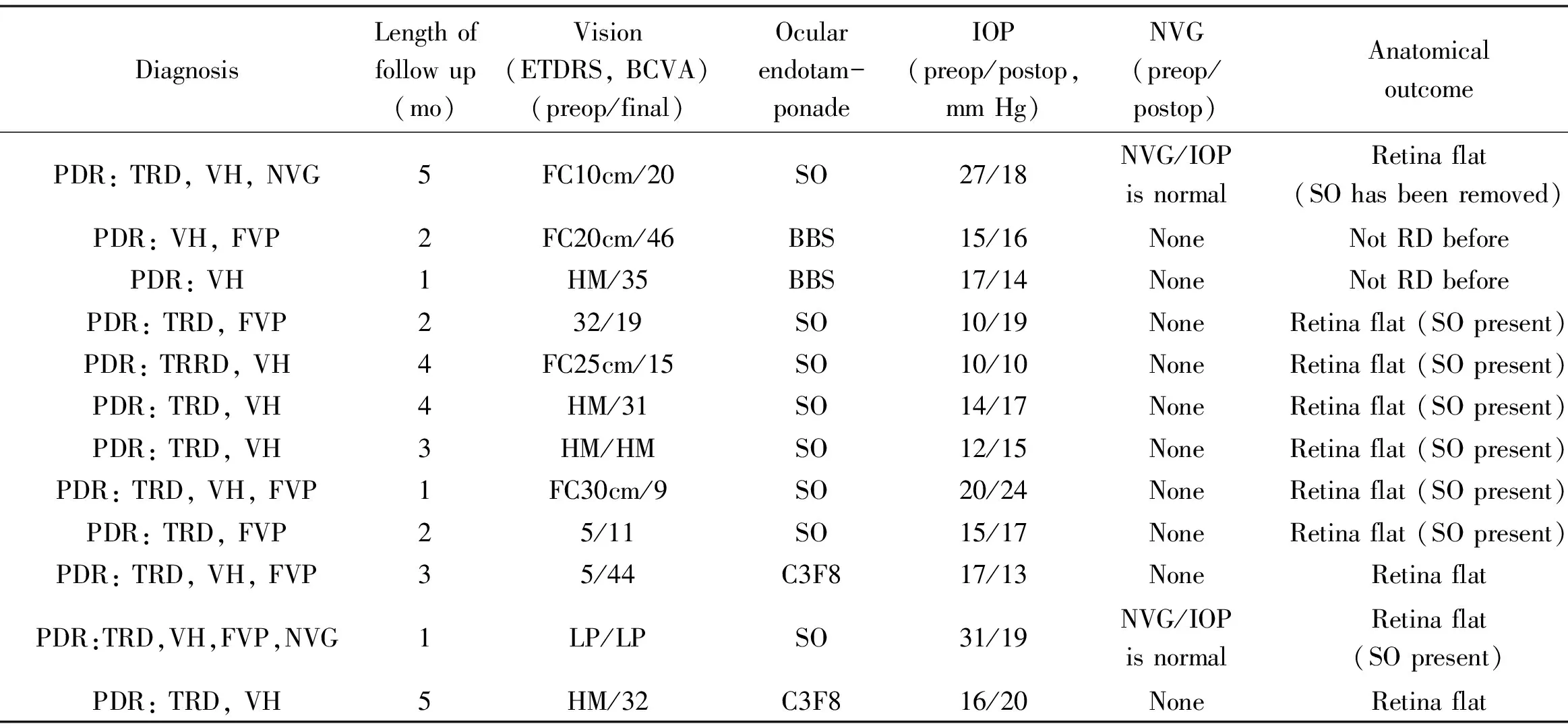
CaseNumberAge/sex/eyeTypeofDMDiagnosisLengthoffollowup(mo)Vision(ETDRS,BCVA)(preop/final)Ocularendotam-ponadeIOP(preop/postop,mmHg)NVG(preop/postop)AnatomicaloutcomeHYP(postop)VH(postop)DME(OCT,postop)143/F/LENIDDMPDR:TRD,VH,NVG5FC10cm/20SO27/18NVG/IOPisnormalRetinaflat(SOhasbeenremoved)NoneNoneNone247/F/LENIDDMPDR:VH,FVP2FC20cm/46BBS15/16NoneNotRDbeforeNoneNoneExist346/M/RENIDDMPDR:VH1HM/35BBS17/14NoneNotRDbeforeNoneNoneNone452/F/LENIDDMPDR:TRD,FVP232/19SO10/19NoneRetinaflat(SOpresent)NoneNoneExist566/M/RENIDDMPDR:TRRD,VH4FC25cm/15SO10/10NoneRetinaflat(SOpresent)NoneNoneNone644/M/RENIDDMPDR:TRD,VH4HM/31SO14/17NoneRetinaflat(SOpresent)NoneNoneNone760/M/RENIDDMPDR:TRD,VH3HM/HMSO12/15NoneRetinaflat(SOpresent)NoneNoneNone850/F/LENIDDMPDR:TRD,VH,FVP1FC30cm/9SO20/24NoneRetinaflat(SOpresent)NoneNoneNone949/F/RENIDDMPDR:TRD,FVP25/11SO15/17NoneRetinaflat(SOpresent)NoneNoneExist1066/F/RENIDDMPDR:TRD,VH,FVP35/44C3F817/13NoneRetinaflatNoneNoneNone1149/F/LENIDDMPDR:TRD,VH,FVP,NVG1LP/LPSO31/19NVG/IOPisnormalRetinaflat(SOpresent)ExistNoneExist1252/M/RENIDDMPDR:TRD,VH5HM/32C3F816/20NoneRetinaflatNoneNoneNone
PDR: Proliferative diabetic retinopathy; PPV: Pars plana vitrectomy; NIDDM: Non insulin dependent diabetes mellitus; PDR: Proliferative diabetic retinopathy; TRRD: Tractional rhegmatogenous retinal detachment; TRD: Tractional retinal detachment; VH: Vitreous Haemorrhage; NVG: Neovascular glaucoma; FVP: Fibrovascular proliferation; HYP : Hyphaema; SO: Silicone oil; BBS: Balanced salt solution; DME: Diabetic macular edema; ETDRS: Early treatment of diabetic retinopathy study; FC: Finger counting; HM: Hand motions; LP: Light projection.
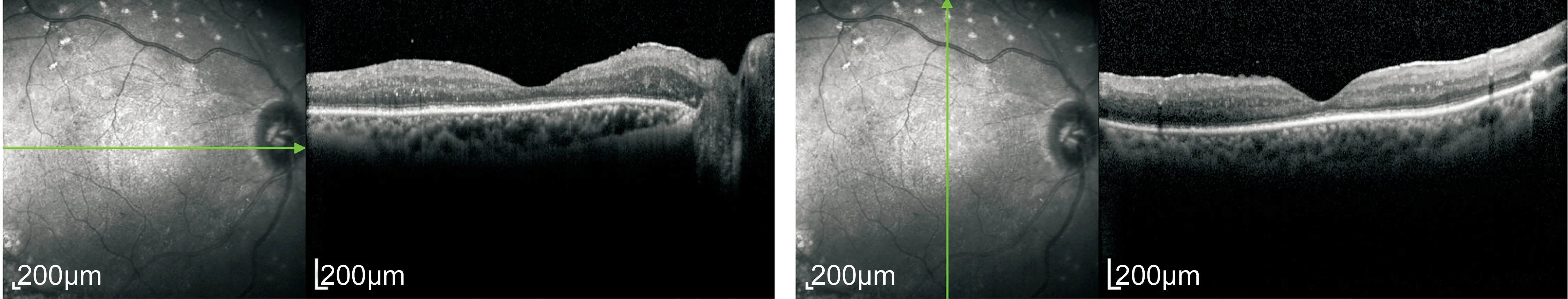
Figure 6 Postoperative optical coherence tomography demonstrates the macula devoid of diabetic macular edema (the same patient with Figure 4).
Postoperatively, the 2 eyes had regressed rubeosis. The IOP of these 2 NVG eyes became normal level after cyclocryotherapy and cyclophotocoagulation.
DISCUSSION
VEGF is a major causative factor in eye diseases characterized by neovascularization or increased vascular permeability such as diabetic retinopathy[3]. VEGF levels in ocular tissues from patients with diabetes are greater than in non-diabetic subjects[4]. Vitreous samples from patients with PDR have elevated VEGF concentrations that are reduced following successful panretinal photocoagulation (PRP)[5]. So, VEGF is the important factor in DR.
As we know, DR is recognized as an inflammatory process, and VEGF has been identified as an important inflammatory mediator. VEGF causes increased expression of the intercellular adhesion molecule-1 (ICAM-1)[6-7]. Leukostasis has been found to correlate temporally with leakage and VEGF could promotes leukostasis[8]. Leukocytes are particularly abundant at the leading edge of areas of pathologic neovascularization[9]. ICAM-1 expression and leukocyte counts are also increased in human diabetic retinas[10]. All these findings provide additional rationale for clinical therapy of VEGF blockade for DR. So, many anti-VEGF drugs (pegaptanib, bevacizumab, ranibizumab, aflibercept and so on) produce an inhibition of VEGF, could improve the vision of people with PDR.
Usually, preoperative intravitreal anti-VEGF drugs were used in PDR therapy. Zamanetal[11]. and colleagues demonstrated that preoperative intravitreal bevacizumab is helpful in improving BCVA post operatively, reducing the time of vitrectomy, decreasing the incidence of intraoperative and postoperative bleeding and reducing the frequency of rubeosis and hyphaema in PDR patients. Ribeiroetal[12]showed that intravitreal ranibizumab could reduce intraoperative bleeding during pars plana vitrectomy for diabetic traction retinal detachment. Intravitreal anti-VEGF before the PPV could increase postoperative BCVA, decrease duration of surgery, reduce intraoperative surgery complications, increase successful rate, and so on[11-13]. But the time taken for anti-VEGF to exert its anti-angiogenic effects sufficient to facilitate surgery is unclear. In our study, all the eyes underwent PPV and SO, C3F8 or BSS tamponade. At the end of the surgery, ranibizumab (Lucentis) has been injected into vitreous cavity. So why we choose this time (at the end of PPV) to inject? Preoperatively, 10 of 12 (83.3%) eyes with VH, we could not observe fundus conditions. So it is not appropriate for intravitreal ranibizumab before the PPV. At the end of the surgery, we could inject ranibizumab to the surface of the posterior pole of the retina under direct vision that is the best place for ranibizumab absorbing (Figure 1). Intravitreal ranibizumab at the end of PPV could reduce the frequence of surgery and decrease the risk of infection. As we metioned before that VEGF has been identified as an important inflammatory mediator in DR. So ranibizumab is more appropriate injected at the end of the PPV for anti-VEGF. In our cases, anatomical attachment was achieved in 10/10 (100%) of the entire RD eyes. Postoperative BCVA improved in 9 eyes (75%). Eight of 12 (66.7%) eyes did not have DME after surgery for 1mo from the OCT examination. The outcomes were so encouraging.
To our knowledge, there are only few case reports on intravitreal anti-VEGF in vitrectomized eyes (with SO tamponade)[14-16]. Falavarjanietal[16]demonstrated that 2.5 mg bevacizumab were injected into human eyes, which later had iris neovascularization (INV) under a silicone oil tamponade, and all five eyes had improved visual acuity and a regression of INV without retinal toxicity. In our study, the BCVA of most eyes were improved, without significant rise of IOP, and without other significant postoperative complications (such as VH, hyphaema and so on). Eight of 12 eyes did not have DME after surgery. So we suggested that 0.5 mg ranibizumab injected into vitrectomized eyes (with SO, C3F8 or BSS tamponade) did not have retinal toxicity. Xuetal[17]observed that following an intra-silicone oil injection of bevacizumab in rabbit’s eyes, the visible bevacizumab droplets migrated in silicone oil for 24-72h before integrating into the vitreous fluid existing between the silicone oil body and retina surface. But in our study, as we mention before, we could inject ranibizumab to the surface of the posterior pole of the retina under direct vision, so the ranibizumab droplets did not need to migrate through the vitreous cavity (filled with SO, C3F8 or BSS), and presence of SO or C3F8 in the vitreous cavity may reduce drug diffusion, increasing drug exposure to the retina and treatment success.
Our results suggest that intravitreal injections of 0.5 mg ranibizumab into vitrectomized eyes at the end of the PPV, is the appropriate time for injection, and may lead to improvement in both functional (BCVA) and morphologic (OCT) parameters of PDR patients, without significant postoperative complications, in vitrectomized eyes (especially for SO tamponade).
1 Randomized clinical trial evaluating intravitreal ranibizumab or saline for vitreous hemorrhage from proliferative diabetic retinopathy.JAMAOphthalmol2013;131(3):283-293
2 Nicholson BP, Schachat AP. A review of clinical trials of anti-VEGF agents for diabetic retinopathy.GraefesArchClinExpOphthalmol2010;248(7):915-930
3 Adamis AP, Miller JW, Bernal MT, D’Amico DJ, Folkman J, Yeo TK, Yeo KT. Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy.AmJOphthalmol1994;118(4):445-450
4 Lutty GA, McLeod DS, Merges C, Diggs A, Plouét J. Localization of vascular endothelial growth factor in human retina and choroid.ArchOphthalmol1996;114(8):971-977
5 Aiello LP, Avery RL, Arrigg PG, Keyt BA, Jampel HD, Shah ST, Pasquale LR, Thieme H, Iwamoto MA, Park JE. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders.NEnglJMed1994;331(22):1480-1487
6 Lu M, Perez VL, Ma N, Miyamoto K, Peng HB, Liao JK, Adamis AP. VEGF increases retinal vascular ICAM-1 expression in vivo.InvestOphthalmolVisSci1999;40(8):1808-1812
7 Barleon B, Sozzani S, Zhou D, Weich HA, Mantovani A, Marmé D. Migration of human monocytes in response to vascular endothelial growth factor (VEGF) is mediated via the VEGF receptor flt-1.Blood1996;87(8):3336-3343
8 Ishida S, Usui T, Yamashiro K, Kaji Y, Ahmed E, Carrasquillo KG, Amano S, Hida T, Oguchi Y, Adamis AP. VEGF164 is proinflammatory in the diabetic retina.InvestOphthalmolVisSci2003;44(5):2155-2162
9 Ishida S, Usui T, Yamashiro K, Kaji Y, Amano S, Ogura Y, Hida T, Oguchi Y, Ambati J, Miller JW, Gragoudas ES, Ng YS, D’Amore PA, Shima DT, Adamis AP. VEGF164-mediated inflammation is required for pathological, but not physiological, ischemia-induced retinal neovascularization.JExpMed2003;198(3):483-489
10 McLeod DS, Lefer DJ, Merges C, Lutty GA. Enhanced expression of intracellular adhesion molecule-1 and P-selectin in the diabetic human retina and choroid.AmJPathol1995;147(3):642-653
11 Zaman Y, Rehman AU, Memon AF. Intravitreal Avastin as an adjunct in patients with proliferative diabetic retinopathy undergoing pars plana vitrectomy.PakJMedSci2013;29(2):590-592
12 Ribeiro JA, Messias A, de Almeida FP, Costa RA, Scott IU, de Figueiredo-Pontes LL, Jorge R. The effect of intravitreal ranibizumab on intraoperative bleeding during pars plana vitrectomy for diabetic traction retinal detachment.BrJOphthalmol2011;95(9):1337-1339
13 Rizzo S, Genovesi-Ebert F, Di Bartolo E, Vento A, Miniaci S, Williams G. Injection of intravitreal bevacizumab (Avastin) as a preoperative adjunct before vitrectomy surgery in the treatment of severe proliferative diabetic retinopathy (PDR).GraefesArchClinExpOphthalmol2008;246(6):837-842
14 Tano R, Kakurai K, Sakurai T, Fujiwara R, Mano T, Maeno T. Intravitreal bevacizumab (avastin) combined with vitrectomy for recurrences of proliferative vitreoretinopathy in von Hippel-Lindau disease.ActaOphthalmologica2011;90(2):e157-e158
15 Cascavilla ML, Querques G, Querques L, Codenotti M, Bandello F. Intravitreal Ranibizumab for myopic choroidal neovascularization after pars plana vitrectomy and silicone oil tamponade.EurJOphthalmol2013;23(6):913-916
16 Falavarjani KG, Modarres M, Nazari H. Therapeutic effect of bevacizumab injected into the silicone oil in eyes with neovascular glaucoma after vitrectomy for advanced diabetic retinopathy.Eye(Lond) 2010;24(4):717-719
17 Xu Y, You Y, Du W, Zhao C, Li J, Mao J, Chen H, Cheng L. Ocular pharmacokinetics of bevacizumab in vitrectomized eyes with silicone oil tamponade.InvestOphthalmolVisSci2012;53(9):5221-5226
严重PDR玻璃体切割术后雷珠单抗注射的效果观察
陈 博,张 宪,杨华静,陈志祺,王帅帅,尹铁梅,孙旭芳
(作者单位:430030 中国湖北省武汉市,华中科技大学同济医学院附属同济医院眼科)
陈博,毕业于华中科技大学同济医学院,博士,主治医师,讲师,研究方向:视网膜病。
孙旭芳,毕业于华中科技大学同济医学院,博士,主任医师,教授,研究方向:视网膜病. dr_sunxf@aliyun.com
目的:对严重增殖性糖尿病视网膜病变的患者行玻璃体切割术后行雷珠单抗注射的效果观察。方法:回归性分析。12例严重增殖性糖尿病视网膜病变患者(12眼)接受睫状体平坦部玻璃体切割术,同时给予硅油、惰性气体或者平衡液的玻璃体腔填充。在手术结束的同时给予雷珠单抗的玻璃体腔注射。结果:随访时间平均为2.75mo。这12眼中分别包括玻璃体积血(1眼);玻璃体积血伴纤维血管化增生(1眼);玻璃体积血伴牵拉性视网膜脱离(3眼);纤维血管化增生伴牵拉性视网膜脱离(2眼);玻璃体积血伴新生血管性青光眼伴牵拉性视网膜脱离(1眼);玻璃体积血伴纤维血管化增生伴牵拉性视网膜脱离(2眼);玻璃体积血伴纤维血管化增生伴新生血管性青光眼伴牵拉性视网膜脱离(1眼);玻璃体积血伴牵拉性孔源性视网膜脱离(1眼)。12眼中,8眼行玻璃体腔硅油填充,2眼行惰性气体填充,2眼行平衡液填充。所有的患者之前均未接受任何治疗。视网膜脱离复位率为10/10(100%)。1眼术后出现前房积血。9眼术后最佳矫正视力较术前提高,2眼无明显变化,1眼较术前下降。OCT检查显示8眼术后未见黄斑水肿。结论:玻璃体切割术后雷珠单抗注射对严重增殖性糖尿病视网膜病变患者有明显的治疗效果:手术成功率明显提高;患者视力显著提高;糖尿病黄斑水肿的发生概率减少;术中及术后并发症的发生率降低。
糖尿病视网膜病变;玻璃体切割术;雷珠单抗
Xu-Fang Sun, Department of Ophthalmology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, Hubei Province, China. dr_sunxf@aliyun.com
10.3980/j.issn.1672-5123.2016.12.02
:Chen B, Zhang X, Yang HJ, Chen ZQ, Wang SS, Yin TM, Sun XF. The effect of intravitreal ranibizumab after pars plana vitrectomy in severe proliferative diabetic retinopathy.GuojiYankeZazhi(IntEyeSci) 2016;16(12):2176-2179
引用:陈博,张宪,杨华静,等. 严重PDR玻璃体切割术后雷珠单抗注射的效果观察.国际眼科杂志2016;16(12):2176-2179

