Preserved egg white alleviates DSS-induced colitis in mice through the reduction of oxidative stress, modulation of inf lammatory cytokines,NF-κB, MAPK and gut microbiota composition
Lingyu Li, Ning Qiu,*, Yqi Meng, Chenyn Wng, Yoshinori Mine, Russell Kest, Vinent Guyonnet
a Key Laboratory of Environment Correlative Dietology, Ministry of Education, College of Food Science and Technology, Huazhong Agricultural University, Wuhan 430070, China
b Department of Food Science, University of Guelph, Guelph, Ontario N1G 2W1, Canada
c CASS Food Research Centre, School of Exercise and Nutrition Sciences, Deakin University, Burwood, Victoria 3125, Australia
d FFI Consulting Ltd, 2488 Lyn Road, Brockville ON K6V 5T3, Canada
Keywords:Preserved egg white Colitis Oxidative stress NF-κB MAPK Gut microbiota
A B S T R A C T Traditional Chinese preserved egg products have exhibited some anti-inflammatory effects, but their mechanisms of action remain unknown. This study aimed to investigate the anti-inflammatory effects of preserved egg white (PEW) treatment on dextran sulfate sodium (DSS)-induced colitis in mice and the underlying mechanisms. The results showed that treatment with PEW in mice with DSS-induced colitis for 14 days effectively improved the clinical signs, inhibited the secretion and gene expression of proinf lammatory cytokines, and reduced myeloperoxidase (MPO) activity and oxidative stress levels. In addition,western blotting results showed that PEW signif icantly suppressed DSS-induced phosphorylation levels of nuclear factor-kappa B (NF-κB) p65 and p38 mitogen-activated protein kinase (MAPK) in colon tissues of mice with colitis. PEW also enhanced the production of short-chain fatty acids (SCFAs) and modulated gut microbiota composition in mice with DSS-induced colitis, including increasing the relative abundance of beneficial bacteria Lachnospiraceae, Ruminococcaceae and Muribaculaceae, and reducing the relative abundance of harmful bacteria Proteobacteria. Taken together, our study demonstrated that preserved egg white could alleviate DSS-induced colitis in mice through the reduction of oxidative stress, modulation of inf lammatory cytokines, NF-κB, MAPK and gut microbiota composition.
1. Introduction
Ulcerative colitis (UC) is a primary form of the inflammatory bowel disease (IBD), which is mainly characterized by the inf lammation of the intestinal mucosa [1]. IBD is usually accompanied by specif ic clinical signs such as persistent abdominal pain, diarrhea,rectal bleeding and severe weight loss [2]. Although the exact etiology of IBD remains unclear, IBD may be the result of a combination of microbial factors, intestinal mucosal barrier dysfunction, oxidative stress, and increased inf lammatory mediators [3,4]. Currently, drugs such as 5-aminosalicylates, corticosteroids, immunosuppressants and antibiotics are commonly used in the treatment of UC, but these drugs show limited eff icacy and may cause adverse reactions, such as hypertension and diabetes [5,6]. Therefore, there is an urgent need to f ind more effective and safer treatment strategies.
Emerging research has revealed that the alteration of the gut microbiota plays a crucial role in the severity and progression of colitis [7,8]. The gut microbiota can form a barrier on the mucosa,reduce the intestinal permeability, and enhance the defense mechanism of the mucosal epithelium [9]. In addition, SCFAs produced by the gut microbiota can prevent neutrophils and macrophages from releasing interleukin (IL)-8, tumor necrosis factor-α (TNF-α) and other inflammatory cytokines by inhibiting the NF-κB pathway, thereby inhibiting inflammation [10]. Meanwhile, the imbalance of the gut microbiota will lead to an increase of harmful bacteria in the intestinal tract and aggravate intestinal inflammation [11,12]. Our previous study has demonstrated that dietary PEW reduced the relative abundance of Proteobacteria, a pathogenic bacterium producing endotoxins thereby contributing to improved intestinal health [13].Also, the administration of certain probiotic strains may reduce intestinal inflammation and damage, improve the intestinal microbial ecology, and prevent the development of colitis [14,15].
Studies have shown that food-derived proteins effectively alleviate the progression of IBD [16,17]. Dietary interventions in DSS-induced colitis mice improved the intestinal barrier, reduced oxidative stress,regulated the inflammatory cytokines as well as the gut microbiota and restored the production of SCFAs in the cecum [18-20].In recent years, many studies have confirmed that proteins,peptides and amino acids derived from avian eggs showed various bioactivities, including anti-microbial, anti-oxidation and antiinflammatory activities [21-23]. For example, ovotransferrin and the peptide derived from ovotransferrin had antioxidant activity, that could scavenge superoxide anions and relieve oxidative stress [24,25].In addition, ovotransferrin was able to alleviate DSS-induced colitis in mice [16]. Preserved egg white is rich in high-quality protein. And the proteins in preserved egg white will mostly be degraded into peptides and free amino acids during the alkaline-pickled process and the digestion in the gastrointestinal tract [26]. Zhang et al. [27,28]reported that four peptides derived from the simulated gastrointestinal digestion of preserved egg white (SGD-PEW) were able to alleviate the progress of inflammation in mice with DSS-induced colitis, and at the same time could inhibit the activation of NF-κB and MAPK in TNF-α-induced Caco-2 cells, thereby alleviating the progress of inflammation. However, the potential mechanism for the protective effect of PEW treatment on UC and whether it can regulate the gut microbiota are still poorly understood.
Therefore, this study aimed to investigate the anti-inflammatory effects of a PEW diet on DSS-induced colitis in mice. The severity of colitis, MPO activity, expression levels of cytokines, oxidative stress levels, gut microbiota composition and SCFAs content were analyzed to clarify the underlying mechanisms involved.
2. Materials and methods
2.1 Animals, diet and experimental design
According to the National Guidelines for Experimental Animal Welfare, the protocols for the animal experiments were authorized by the Ethics Committee of the Experimental Animal Center of Huazhong Agricultural University (Permission No. HZAUMO-2020-0058).Thirty specific pathogen free (SPF) female BALB/c mice (6–8 weeks of age, 16–18 g) were obtained and housed in the Experimental Animal Center of Huazhong Agricultural University (Wuhan, China).The mice were kept in a SPF experimental animal room with a humidity of (50 ± 5)%, at (21 ± 1) °C, 12 h light/dark cycle, allowing unrestricted access to food and water for one week of adjustable feeding. The mice were randomly divided into three different groups(n= 10) as follows: the normal control group , the DSS-induced colitis group (DSS), and the preserved egg white group (PEW). Mice in the PEW group were given by oral gavage PEW (860 mg/(kg·day))dissolved in saline for 14 days while the other 2 groups of mice received normal saline (0.2 mL per mouse per day) by oral gavage during the same period. Starting on the eighth day, mice in the DSS and PEW groups were given 4% DSS (molecular weight 36 000–50 000, MP Biomedicals, LLC, Irvine, CA) in the drinking water for 7 days, with the DSS solution changed every other day.
The preserved eggs were obtained from Hubei Shendan Healthy Food Co., Ltd. (Hubei, China). After stripping the cleaned preserved eggs of the shell and membranes, the preserved egg white portion was grinded and freeze-dried. The freeze-dried preserved egg white powder was stored at –20 °C until use [13].
2.2 Evaluation of the severity of coliti
The disease activity index (DAI) was determined as synthetical scores of weight loss, stool consistency, and rectal bleeding divided by 3. During the experimental period, the changes of the body weight and the DAI of each mouse were recorded daily according to the method of the previous literature [29]. After 14 days, the mice were euthanized and for each mouse, the colon was excised between the ileo-cecal junction and the proximal rectum and its length was measured and recorded immediately. After these measurements, the cecal samples were cut longitudinally and the fresh contents were collected, placed in sterile tubes and stored at –80 °C for microbiota analysis. The colon tissues were washed with pre-chilled phosphatebuffered saline (PBS, pH 7.4), cut into two parts and the distal colon portions were fixed in 4% paraformaldehyde for histopathological analysis. In order to quantitatively evaluate the severity of the pathological change, histological analysis was assessed according to the previously established grading system (Table S1) [28-30].The proximal portion of the colons were stored at -80 °C to assess later the activities of superoxide dismutase (SOD), malondialdehyde(MDA), glutathione peroxidase (GSH-Px), MPO, nitric oxide (NO)and inflammatory cytokines.
2.3 Measurements of NO, MPO, MDA, GSH-Px, SOD activities and cytokines levels in colon tissues
After thawing, the colon tissues were weighed and a 10% tissue homogenate was prepared with pre-chilled PBS (0.01 mol/L, pH 7.4).The suspensions were centrifuged at 3 000 r/min for 10 min, after which the supernatants were collected for testing. The MPO,MDA, SOD and GSH-Px activities as well as the NO index in the colon tissue were evaluated with the corresponding kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s instructions. The levels of cytokines (TNF-α, IL-6,IL-1β and IL-10) in mice colon tissues were determined using enzyme-linked immunosorbent assay (ELISA) kits (Elabscience,Wuhan, China). The measurement of the optical density of the colorimetric reaction was performed at 450 nm using a flat microplate reader (Molecular Devices, Silicon Valley, USA).
2.4 Analysis of relative mRNA expression in colon tissues
Total RNA was isolated from colon tissues using the TRIZOL®Reagent (Thermo Fisher Scientific, MA, USA). A real-time polymerase chain reaction (PCR) Sequence Detection System(Applied Biosystems, CA, USA) was used to perform quantitative real-time polymerase chain reaction (qRT-PCR) using SYBR Green PCR Master Mix according to the manufacturer’s protocol (Thermo Fisher Scientific). The relative gene expression was calculated using the 2−ΔΔCtmethod and the target gene expression was normalized using the expression of the glyceraldehyde-3-phosphate dehydrogenase(GAPDH) gene [28]. The sequences of the primers used in this study are listed in Table S2.
2.5 Western blot analysis
To extract proteins, the colon tissues were lysed and homogenized in an ice-cold RIPA buffer (containing PMSF and phosphatase inhibitors) and centrifuged at 12 000 r/min at 4 °C for 5 min. The supernatant was then collected for protein quantification using the BCA protein assay kits (Beyotime Biotechnology, Shanghai, China).The protein samples were separated by 10% sodium dodecyl sulfatepolyacrylamide gel electrophoresis (SDS-PAGE) and then transferred onto a polyvinylidene difluoride (PVDF) membrane (Millipore, MA,USA). The PVDF membrane was soaked for 2 h at room temperature in a TBST (blocking solution) shaker containing 5% nonfat milk, then incubated overnight at 4 °C with primary antibodies. The membrane was washed with TBST and then incubated at room temperature for 2 h with HRP-conjugated diluted secondary antibodies. Finally, the proteins bands were visualized with an enhanced chemiluminiscence imaging system (Bio-Rad Laboratories, CA, USA) and results were quantified using the ImageJ software.
2.6 Determination of SCFAs in cecal samples
Cecal samples were collected directly after the mice were euthanized and stored at -80 °C until analysis. The quantitative analysis of SCFAs in the cecal samples was determined by gas chromatography (GC). Briefly, 100 mg of the cecal samples were added into a 2 mL grinding tube with grinding beads and 1 mL of distilled water, containing 0.5% phosphoric acid and 50 μg/mL 2-ethylbutyric acid. The samples were then placed in a cryogrinder and ground twice for 3 min. The mixture was centrifuged at 13 000 ×gat 4 °C for 10 min. The supernatant was mixed with 500 μL of ethyl acetate for extraction and then centrifuged again (13 000 ×g,4 °C, 10 min). The concentrations of the SCFAs were evaluated with a HP-FFAP capillary column (30 m × 0.25 mm × 0.25 μm, Agilent J&W Scientific, Folsom, CA, USA).
2.7 DNA extraction and high-throughput sequencing of cecal samples
Total DNA was extracted from the cecal samples using the QIAamp DNA Stool Mini Kit (Qiagen, Doncaster, Australia).The extracted genomic DNA was placed in the 1% agarose gel electrophoresis for qualitative and quantitative evaluations. Universal primers 338F (5′-ACTCCTACGGGAGGCAGCAG-3′) and 806R(5′-GGACTACHVGGGTWTCTAAT-3′) were used to amplify the V3-V4 region of the bacterial 16S rRNA gene. Sequencing was performed using the Illumina Miseq platform (Illumina, San Diego,California, USA).
2.8 Bioinformatics and statistical analysis
The raw sequencing data were stored in the FASTQ format.The Trimmomatic software was used for quality filtering. The paired-end reads were then merged using the Flash software(version 1.2.11, https://ccb.jhu.edu/software/FLASH/index.shtml).The assembly parameters were as follows: 10 bp of minimal overlap, 200 bp of the maximum overlap, and 20% maximum mismatch rate. The default parameters in the Quantitative Insights into Microbial Ecology (QIIME) software (version 1.9.1, http://qiime.org/install/index.html) were set to delete some readings for quality filtering. Operational taxonomic units (OTUs) clustering of non-repetitive sequences (without single sequences) was performed using the Usearch software (version 7.0, http://www.drive5.com/usearch/), and chimeras in reads were detected and removed in the clustering process to generate OTUs with a 97% similarity cutoff.All representative reads were annotated and blasted against the SILVA database Version 138 (16S rDNA) using the RDP classifier(confidence threshold was 70%) [31].
All results were expressed as mean ± standard deviation (SD). The statistical significance of differences between groups was analyzed by one-way analysis of variance (ANOVA) and Duncan’s multiple range tests using SPSS (Version 21.0, IBM, USA). Origin Pro 9.0 was used for graphical processing.
3. Results
3.1 Dietary PEW alleviated the clinical signs of DSS-induced colitis in mice
There was no difference in the average initial weight of mice among the groups. The body weight of the mice in the control group showed a gradual upward trend throughout the experimental period while the DSS and PEW groups also showed an increased body weight only during the first 7 days, as shown in Fig. 1A. After initiating the administration of DSS, the body weight of mice in the DSS and PEW groups decreased, starting on day 11. However, mice in the PEW group had a significantly (P< 0.05) lower reduction in body weight than the mice in the DSS group.
The DAI score is the mean synthetical score of weight loss, stool consistency, and rectal bleeding. The mice in the control group didn’t lose body weight markedly and didn’t show any clinical symptoms,giving the value of 0 in the DAI score. With the increase of DSStreatment days, the DAI scores of the DSS and PEW groups gradually increased, while mice in the PEW group had a significantly lower increase in DAI score than the mice in the DSS group (P< 0.05) (Fig. 1B).Since the shortening of the colon has been identified as one of the main signs in DSS-induced colitis, the length of the colon can be used as an indicator reflecting the severity of inflammation [32].When compared with the control group, the colon length in the DSS group was significantly shortened (P< 0.05) while the administration of PEW significantly limited the shortening of the colon (P< 0.05)(Fig. 1C).
3.2 Dietary PEW improved histological damage of colon tissues in DSS-induced colitis mice
In order to explore the effect of PEW on the severity of inflammation and colon morphology on DSS-induced colitis,pathological examinations were performed on colon sections after hematoxylin-eosin (H&E) staining. As shown in Fig. 1D, the colon tissue sections in the control group mice exhibited normal colon tissue, with intact mucosal epithelium, minimal inflammatory cell infiltration, and no other obvious abnormalities. However, the colon tissues in the DSS group mice displayed localized tissue ulcers, missing mucosal epithelium and intestinal gland structure,submucosal edema, and severe inflammatory cells infiltration in the mucosal and submucosa layers, resulting in higher histological scores. In contrast to the DSS group, mice treated with PEW showed less crypt shortening and reduced mucosal damage along with submucosal inflammatory cells infiltration, resulting in significantly lower histological scores (P< 0.05). These results indicated that PEW treatment alleviated the inflammation of the colon.
3.3 Dietary PEW reduced MPO activity and oxidative stress levels in the colon tissues of mice with DSS-induced colitis
MPO is a pro-inflammatory and pro-oxidant enzyme that can cause colon tissues damage [33,34]. Compared with the control group, the MPO activity in the colonic tissues of the DSS group was significantly increased (P< 0.05), while the PEW treatment significantly reduced the MPO level (P< 0.05) (Fig. 2A). In order to evaluate the effect of PEW treatment on colonic oxidative stress, we measured MDA level, SOD activity, GSH-Px activity and NO level. Compared with the control group, the MDA (Fig. 2B) and NO (Fig. 2E)levels in the colon tissues in the DSS group were significantly increased (P< 0.05). Treatment with PEW significantly reduced the MDA and NO levels compared with the DSS group (P< 0.05). In addition, the SOD (Fig. 2C) and GSH-Px (Fig. 2D) activities in the colonic tissues in the DSS group were significantly reduced (P< 0.05),while the PEW treatment significantly increased the SOD and GSH-Px activity compared with the DSS group (P< 0.05). These results indicated that PEW treatment improved the ability of mice with DSSinduced colitis to scavenge free radicals and inhibit oxidative stress.
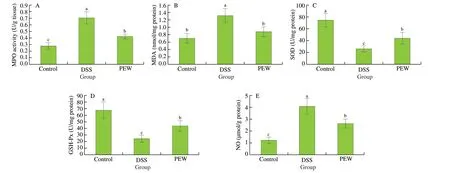
Fig. 2 Dietary PEW reduced MPO activity and oxidative stress levels in the colon tissues of mice with DSS-induced colitis. (A) MPO activity, (B) MDA,(C) SOD, (D) GSH-Px, and (E) NO. Data are presented as mean ± SD (n = 5 per group); Values without a common letter are significantly different at P < 0.05.
3.4 Dietary PEW reduced the secretion of pro-inflammatory cytokines and increased the secretion of anti-inflammatory cytokines in the colon tissues of mice with DSS-induced colitis
In order to evaluate the influence of PEW on inflammatory cytokines, the concentrations of TNF-α, IL-6, IL-1β and IL-10 in the colon tissues were measured by ELISA. Pro-inflammatory cytokine(including TNF-α, IL-6 and IL-1β) levels in the DSS group mice were significantly higher (P< 0.05) than those in the control group mice(Figs. 3A-C). Compared with the levels observed in the DSS group,the dietary PEW significantly reduced the secretion of all three proinflammatory cytokines (P< 0.05). Concurrently, the level of the antiinflammatory cytokine IL-10 in the colon tissues of the PEW group was significantly increased (P< 0.05) when compared to the DSS group (Fig. 3D).
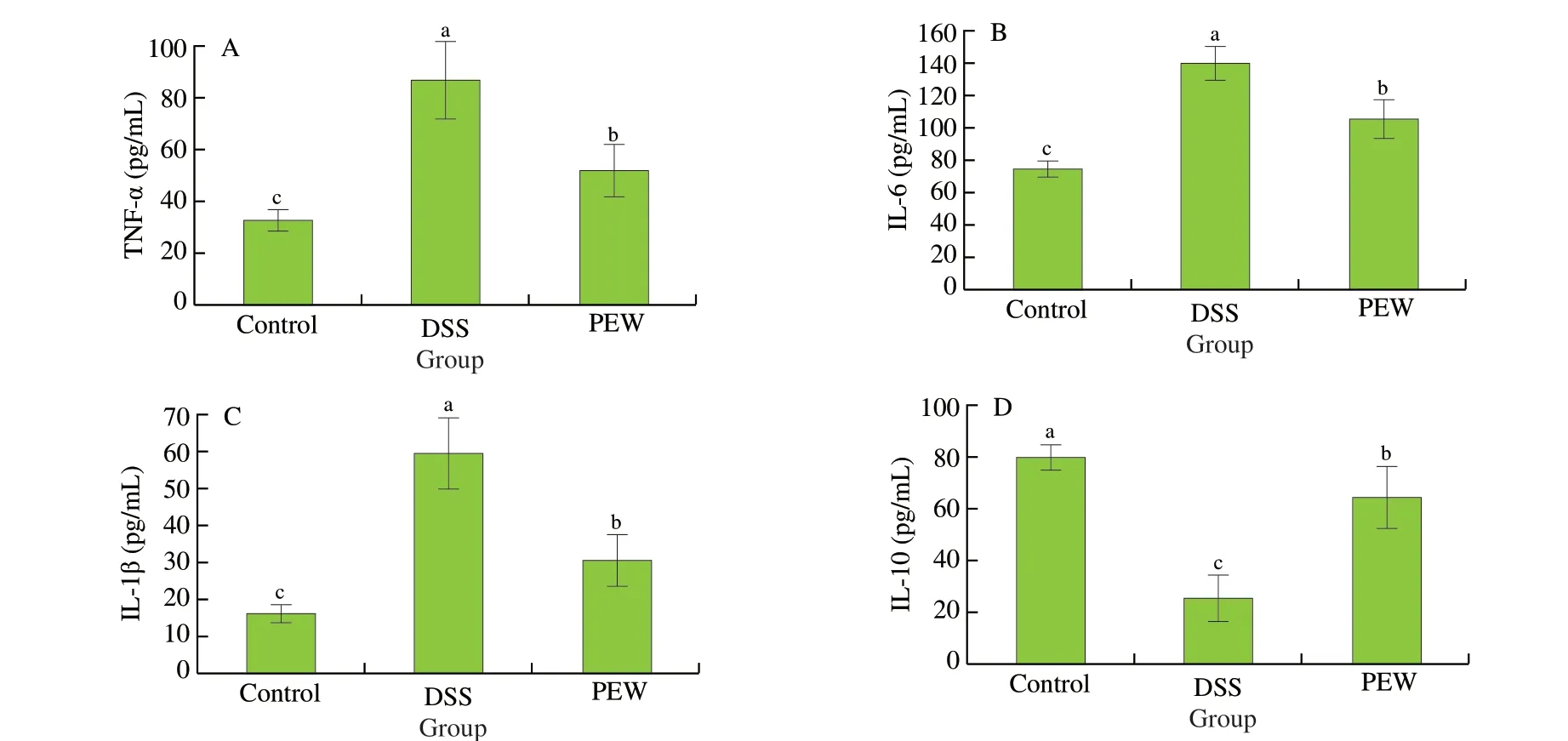
Fig. 3 Dietary PEW reduced the concentrations of pro-inflammatory cytokines TNF-α (A), IL-6 (B), IL-1β (C) and increased the concentration of antiinflammatory cytokines IL-10 (D) in the colon tissues of mice with DSS-induced colitis. Data are presented as mean ± SD (n = 5 per group); Values without a common letter are significantly different at P < 0.05.
3.5 Dietary PEW modulated the gene expression of inflammatory cytokines in the colon tissues of mice with DSSinduced colitis
The anti-inflammatory effect of PEW on DSS-induced colitis was studied by measuring the mRNA expression of inflammatory cytokines in the colon tissues. Compared with the control group,the mRNA expression of pro-inflammatory cytokinesTNF-α,IL-6andIL-1βwere significantly increased (P< 0.05) in the DSS group mice (Figs. 4A-C). PEW treatment significantly reduced the mRNA expression ofTNF-α, IL-6andIL-1βinduced by DSS (P< 0.05). In addition, the mRNA expression of the anti-inflammatory cytokineIL-10in the DSS group was significantly reduced (P< 0.05)when compared with the control group. Dietary PEW significantly increased the mRNA expression ofIL-10(P< 0.05) compared to the DSS group although not to a level comparable to the control group (Fig. 4D).
3.6 Dietary PEW regulated p38 MAPK and NF-κB p65 pathways in the colon tissues of mice with DSS-induced colitis
The phosphorylation of NF-κB p65 and p38 MAPK in colon tissues was examined in order to investigate whether the inhibition of inflammation by PEW was mediated through the NF-κB and MAPK pathways. As shown in Fig. 5A and B, compared with the control group, the phosphorylation levels of NF-κB p65 and p38 MAPK proteins in colon tissues of the DSS group significantly increased(P< 0.05). The PEW treatment significantly reduced (P< 0.05) the phosphorylation levels when compared to the DSS group although not to a level similar to that in the control group.
3.7 Dietary PEW modulated gut microbiota composition in mice with DSS-induced colitis
3.7.1 Diversity analysis of gut microbiota
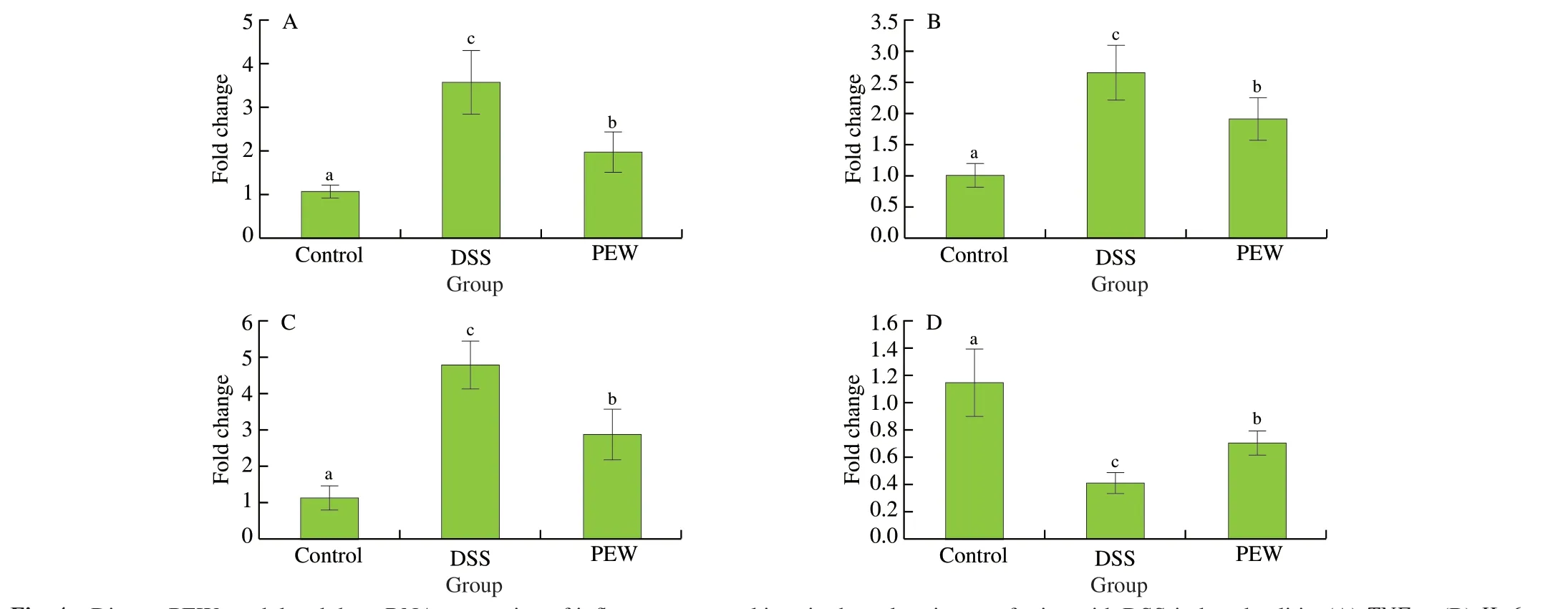
Fig. 4 Dietary PEW modulated the mRNA expression of inflammatory cytokines in the colon tissues of mice with DSS-induced colitis. (A) TNF-α, (B) IL-6,(C) IL-1β, and (D) IL-10. Data are presented as mean ± SD (n = 5 per group); Values without a common letter are significantly different at P < 0.05.
In order to investigate whether PEW could modify the gut microbiota, 16S rRNA gene sequencing analysis was performed to determine the changes in the composition of the gut microbiota among the three groups. Shannon, Simpson and Sobs (observed richness)were used to assess the alpha diversity among groups. Shannon and Simpson indexes are commonly used to evaluate microbial diversity of communities while the Sobs index is mainly used to evaluate the richness of microbes within communities. The Shannon and Sobs indexes of the DSS group (Fig. 6A and C) were significantly lower compared with the control group (P< 0.001), while the Simpson index of the DSS group (Fig. 6B) was significantly higher compared with the control group (P< 0.001). In the PEW treatment, the Shannon and Sobs indexes increased significantly (P< 0.05), while the Simpson index decreased significantly (P< 0.05), indicating PEW treatment promotes the community diversity and richness of the gut microbiota. Beta diversity analysis provides information about the similarity of community structure between different treatment groups. The beta diversity was evaluated by principal coordinate analysis (PCoA) using weighted UniFrac distance. The distance between the points for the control group and the DSS group indicated that the gut microbiota structure in the DSS group deviated from the control group. In the PEW group, the gut microbiota induced by the administration of DSS shifted closer to that of the control group(Fig. 6D), indicating that dietary PEW improved the beta diversity of the gut microbiota of colitis mice.
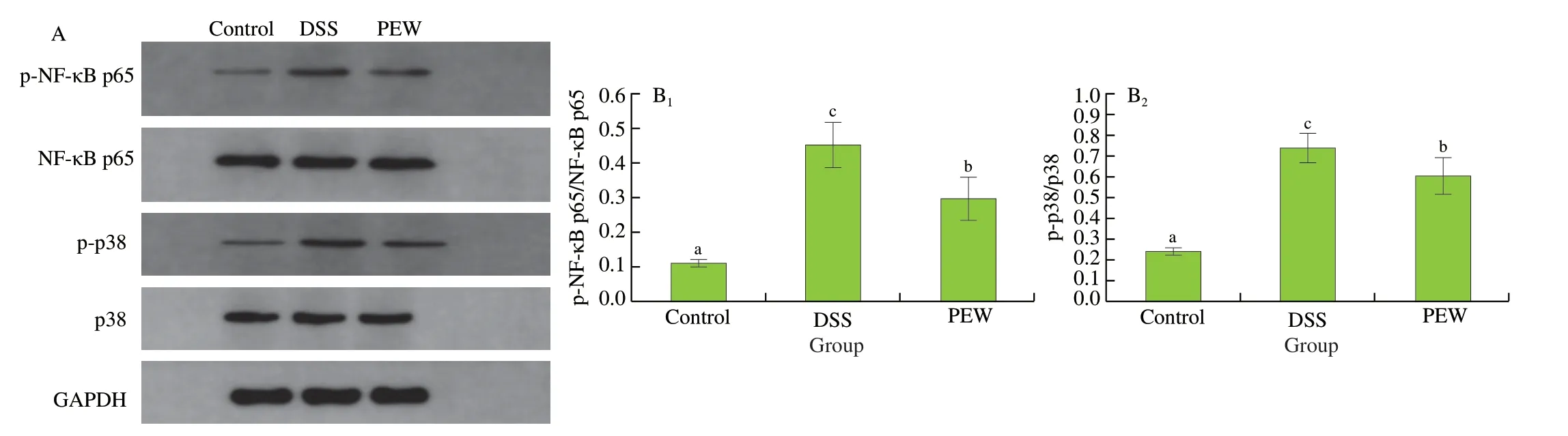
Fig. 5 Dietary PEW regulated the p38 MAPK and NF-κB p65 pathways in the colon tissues of mice with DSS-induced colitis. (A) Representative image of western blotting, (B) protein expression of NF-κB p65 (B1), and p38 MAPK (B2). GAPDH was used as a control. Data are presented as mean ± SD (n = 3 per group); Values without a common letter are significantly different at P < 0.05.
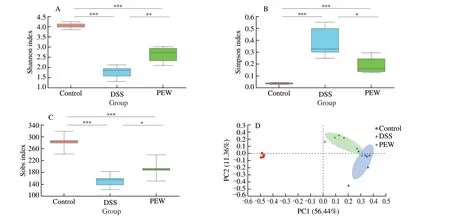
Fig. 6 Dietary PEW improved the alpha and beta diversity of the gut microbiota in colitis mice. (A) Shannon index, (B) Simpson index, (C) Sobs indexes, and(D) principal coordinate analysis (PCoA) using weighted UniFrac distance. Each plot represents one sample, n = 6; * P < 0.05, ** P < 0.01, *** P < 0.001.
3.7.2 Composition and relative abundance of gut microbiota
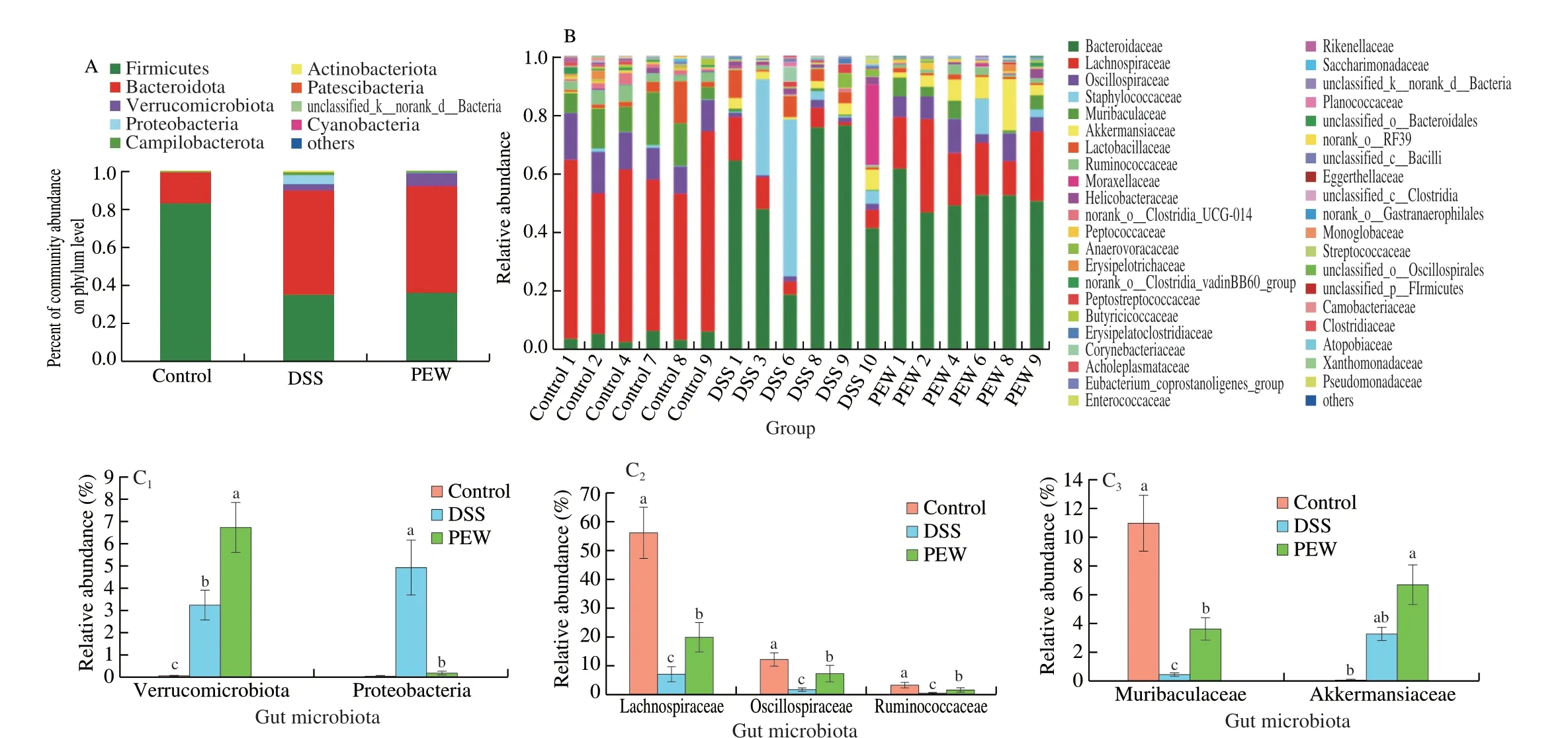
Fig. 7 Dietary PEW modulated the taxonomic composition of the gut microbiota in DSS-induced mice. (A) Relative abundance of the gut microbial community at the phylum level, (B) relative abundance of the gut microbial community at the family level, and (C) the relative abundance of Verrucomicrobiota,Proteobacteria, Muribaculaceae, Akkermansiaceae, Lachnospiraceae, Oscillospiraceae and Ruminococcaceae at the phylum and family levels. Data are presented as mean ± SD (n = 6 per group); Values without a common letter are significantly different at P < 0.05.
Fig. 7A-B separately show the taxonomic composition of the fecal microbiota of mice at the phylum and family levels. Nine types of gut microbiota compositions, including Firmicutes, Bacteroidota,Verrucomicrobiota, Proteobacteria, Campilobacterota and other floras were detected at the phylum level (Fig. 7A). In the control group, the relative abundances of Firmicutes, Bacteroidota, Verrucomicrobiota and Campilobacterota were 82.72%, 15.96%, 0.08% and 0.61%,respectively. The administration of DSS changed the bacterial composition at the phylum level. In the DSS group, the relative abundances of Bacteroidota, Verrucomicrobiota, Proteobacteria and Campilobacterota increased to 54.37%, 3.29%, 4.94% and 1.44%,respectively. Conversely, the relative abundance of Firmicutes decreased to 34.85%. Among all experimental groups, Firmicutes (F)and Bacteroidota (B) were the most important taxa. The ratio of F/B is an important indicator for evaluating microbial changes [35,36]. The ratio of F/B in the DSS group (F/B, 0.641 0) was significantly lower(P< 0.05) than that in the control group (F/B, 5.183 0). Compared to the DSS group, the treatment with PEW increased the relative abundance of Verrucomicrobiota (P< 0.05) and decreased the relative abundance of Proteobacteria (P< 0.05). The changes in the relative abundances of Verrucomicrobiota and Proteobacteria among the three groups were plotted separately (Fig. 7C). Next, we determined the differences at the family level among the treatment groups (Fig. 7B). The dominant floras at the family level were Lachnospiraceae, Oscillospiraceae,Muribaculaceae, Ruminococcaceae and Akkermansiaceae in the three groups. The changes among the three groups in the relative abundances of Lachnospiraceae, Oscillospiraceae, Muribaculaceae, Ruminococcaceae and Akkermansiaceae were plotted separately (Fig. 7C). Compared to the control group, the relative abundances of Lachnospiraceae,Oscillospiraceae, Muribaculaceae, and Ruminococcaceae in the DSS group decreased significantly (P< 0.05). In the PEW group, the relative abundances of these floras increased significantly (P< 0.05)although not to a level comparable to those in the control group.Interestingly, compared with the control group, DSS treatment and PEW treatment increased significantly the relative abundance of Akkermansiaceae (P< 0.05).
Fig. 8A shows the taxonomic composition of the fecal microbiota of mice at the genus levels with the extended error bar plots revealing significant differences of the mean proportions of bacterial taxa between the control and DSS groups (Fig. 8B), the DSS and PEW groups (Fig. 8C) at the genus level. As shown in the Fig. 8A, the taxonomic composition with the higher relative abundance at the genus level mainly includedBacteroides, Lachnospiraceae_NK4A136_group, Staphylococcus, norank_f_Muribaculaceae,unclassified_f_Lachnospiraceae,Blautia,Akkermansia. As shown in Fig. 8B, the disruption of gut microbiota symbiosis in the DSS group was characterized by a higher relative abundance ofBacteroides(P< 0.01) andStaphylococcus(P< 0.05), which have been confirmed to aggravate colitis [18,20], and a lower relative abundance of Lachnospiraceae_NK4A136_group (P< 0.01), norank_f_Muribaculaceae (P< 0.01), unclassified_f_Lachnospiraceae(P< 0.01) andBlautia(P< 0.05) than in the control group. Moreover,dietary PEW increased the relative abundance ofAkkermansia(Fig. 8A),unclassified_f_Lachnospiraceae (P< 0.05) andBlautia(P< 0.05)(Fig. 8C) compared with the DSS group. Considered together, these results indicated that the administration of PEW was able to regulate the composition of the gut microbiota in DSS-induced colitis.
3.8 Dietary PEW promoted the production of SCFAs in the cecum
The content of SCFAs of the cecum of each group of mice was determined by gas chromatography. As shown in Fig. 9, the cecal levels of acetic acid (Fig. 9A), butanoic acid (Fig. 9C) and total short-chain fatty acids (Fig. 9D) in the DSS group were significantly lower than those in the control group (P< 0.05). In the PEW group,the cecal levels of acetic acid, butanoic acid and total short-chain fatty acids increased significantly when compared to the DSS group(P< 0.05), but remained lower than the levels observed in the control group. Interestingly, the cecal level of propionic acid (Fig. 9B) was higher in the PEW group than in the control group.
4. Discussion

Fig. 8 Dietary PEW modulated the taxonomic composition of the gut microbiota in DSS-induced mice. (A) Relative abundance of the gut microbial community at the genus level. Phylotypes between the control and DSS groups (B), the DSS and PEW groups (C) at the genus level were significantly different; the differences between experimental groups were determined using a Wilcoxon rank-sum test, and the FDR (Falsely Discovery Rate) procedure was used to control the falsediscovery rate. In each group, n = 6, * P < 0.05, ** P < 0.01.
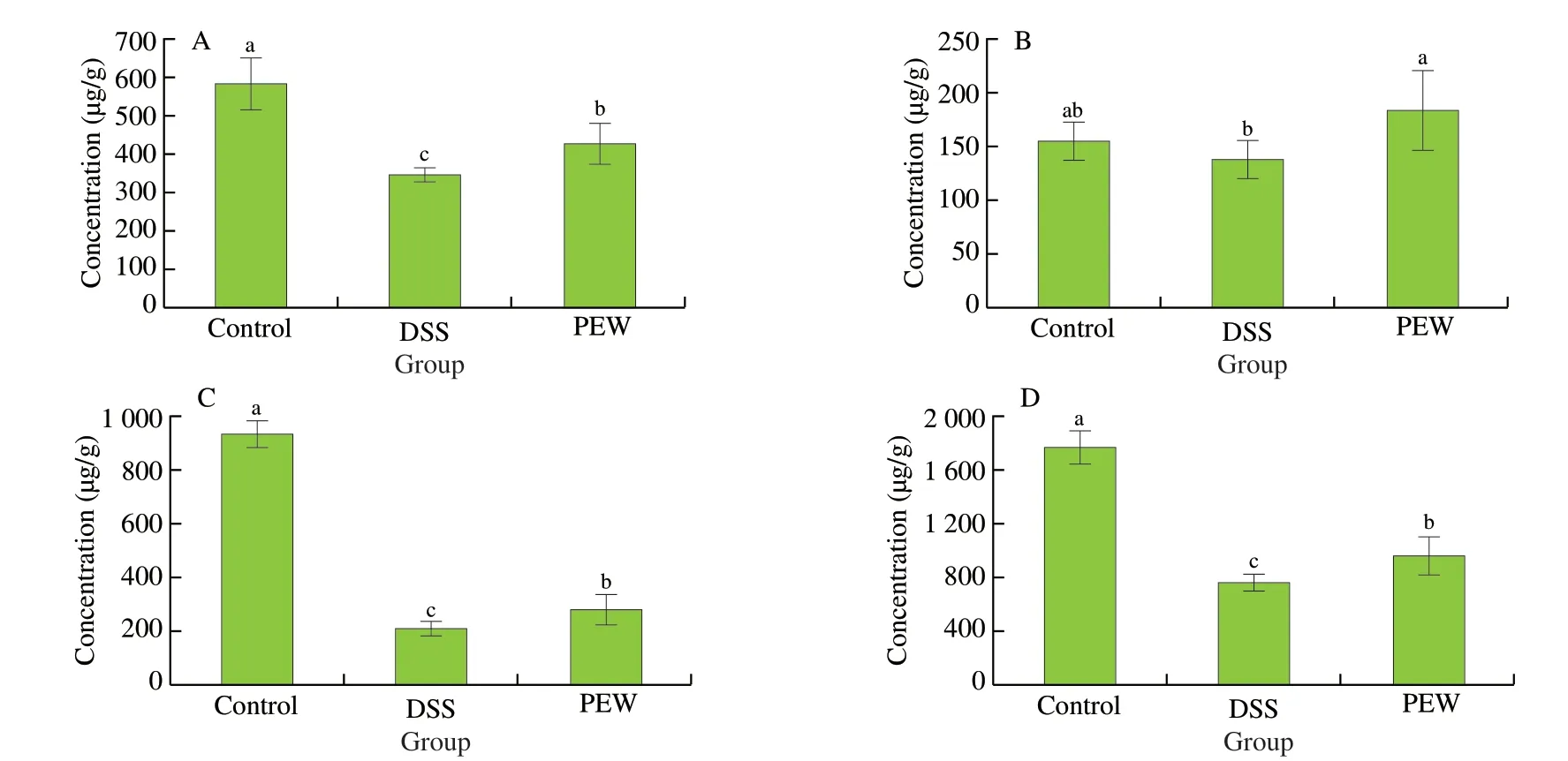
Fig. 9 Dietary PEW promoted the production of SCFAs in the cecum. (A) acetic acid, (B) propionic acid, (C) butanoic acid, and (D) total short-chain fatty acids.Data are presented as mean ± SD (n = 6 per group); Values without a common letter are significantly different at P < 0.05.
IBD, including Crohn’s disease (CD) and UC, is a multifactorial,long-term and incurable disease [3,4]. Pathologies in mice with DSSinduced colitis are typically diarrhea, intestinal bleeding, weight loss, colon shortening, as well as mucosal ulcers and neutrophil infiltration [37,38]. In this study, dietary PEW significantly improved the weight loss, DAI scores, and colon shortening in DSS-treated mice. In addition, the administration of PEW effectively decreased intestinal mucosal damage, submucosal edema, inflammatory cells infiltration and crypt loss. Consequently, mice in the PEW group had significantly reduced histological scores. These results were somewhat similar to a previous report in which the eggshell membrane hydrolysate reduced DSS-induced inflammation in mice [39].Cytokines and their related genes play a vital role in the development and persistence of IBD. The pathophysiological characteristics of UC include the inflammatory cascade and the release of a variety of pro-inflammatory and anti-inflammatory cytokines [40,41]. The overexpression of pro-inflammatory cytokines will cause inflammation of the intestinal mucosa. TNF-α is a key promoter of intestinal epithelial barrier damage in colitis, and it has the ability to promote the production of IL-6 and IL-1β. IL-10 is a major anti-inflammatory cytokine and plays an important role in the maintenance of gastrointestinal homeostasis [11].The pharmacological administration of IL-10 alleviated colitis in mice by suppressing the production of pro-inflammatory cytokines and inhibiting intestinal inflammation [42]. In this study, we proved that dietary PEW significantly reduced the production and gene expression of pro-inflammatory cytokinesTNF-α,IL-6andIL-1βin the colon tissues of mice with colitis, while concurrently increasing the secretion and gene expression ofIL-10in the colon tissues.These results would indicate that dietary PEW alleviated the clinical symptoms in mice with DSS-induced colitis and may reduce the severity of colitis in mice by down-regulating the levels of proinflammatory cytokines in colon tissues.
Various studies have shown that NF-κB and MAPK signaling pathways were activated in the colonic mucosa of UC patients and mice with DSS-induced colitis [43,44]. The activation of these signaling pathways contributes to the development and progression of UC through the phosphorylation of p38 MAPK, NF-κB p65, etc [45].Therefore, we explored the potential effects of PEW on the activation of NF-κB p65 and p38 MAPK. Our results showed that the NF-κB and MAPKs signaling pathways in the colon tissues of DSS-induced colitis mice were activated. Treatment with PEW was able to reduce the phosphorylation of NF-κB p65 and p38 MAPK, in a similar fashion as egg protein ovotransferrin derived peptide IRW was found to exhibit an anti-inflammatory effect by blocking the nuclear translocation of NF-κB p65 [22]. The phosphorylation of NF-κB and MAPK signaling pathways can induce the secretion and the gene expression of considerable amounts of inflammatory cytokines(including TNF-α, IL-6 and IL-1β) and exacerbates the inflammatory response [46]. In our study, after the administration of DSS, there was a significant increase in the mRNA expression of pro-inflammatory cytokinesTNF-α,IL-6andIL-1βin the colon tissues of mice affected with colitis, while the mRNA expression of anti-inflammatory cytokineIL-10decreased. The administration of PEW significantly reduced the mRNA expression of pro-inflammatory cytokines and upregulated the mRNA expression of the anti-inflammatory cytokine in the colon tissues of colitis-affected mice. Therefore, the PEW treatment inhibited the phosphorylation of NF-κB and MAPKs and reduced the secretion and expression of pro-inflammatory cytokines,thereby alleviating intestinal inflammation.
The infiltration of neutrophils into the colon mucosa during UC can secrete a large number of inflammatory cytokines exacerbating the inflammatory state [47]. MPO is an inflammation marker of the infiltration of neutrophils into inflamed and damaged tissues. In the current study, compared with the control group, the MPO activity in the colon tissues of DSS-induced colitis mice was significantly increased (P< 0.05), while the administration of PEW reduced the MPO activity as well as the infiltration of neutrophils in the colon tissues. The activation of inflammatory cells in the intestine can produce more reactive oxygen species, leading to tissue damage and oxidative stress. Oxidative stress is considered as the underlying mechanism in the pathophysiology of IBD, and excessive oxygen radicals are the key factors in the formation of ulcerative inflammatory tissue damage and colitis [48]. The levels of MDA, SOD and GSH-Px levels are good indications of the degree of oxidative stress. We found that after DSS treatment, the activity of SOD and GSH-Px in the colon tissues of mice with colitis decreased while that of MDA increased. Some studies have reported that excessive amount of MDA could induce the production of NO, providing oxygen radicals that led eventually to the infiltration of a large number of inflammatory cells in the colon tissues [12]. We report the level of NO in the colon tissues of mice in the DSS group increased, which was consistent with our observations of a large number of inflammatory cells infiltrating the mucosa of the colon tissues in the DSS group as shown by the H&E staining in Fig. 1D. The administration of PEW inhibited the increase in MDA and NO levels in the colon tissues of mice with DSS-induced colitis while reducing the activity of SOD and GSH-Px. Our results are consistent with other studies where dietary interventions in mice were able to regulate the production of NO, MDA, T-SOD and MPO to repair the oxidative damage of the mucosal barrier and alleviate colitis [47,48]. Our results showed that the treatment with PEW gave the ability to scavenge oxygen radicals and reduce intestinal oxidative stress, therefore alleviating the effects of inflammation.
Previous research has shown that the dysbiosis of the gut microbiota is closely related to the occurrence and development of UC [49]. A germ-free mouse model demonstrated that severe colonic inflammation could not be triggered in the absence of gut microbiota [50].Various reports have indicated that the diversity and abundance of microorganisms decrease in UC patients and DSS-induced animal models [12,15,38]. In our study, PEW treatment increased the diversity and abundance of the gut microbiota when compared to that in the DSS treatment mice.Akkermansiais a gram-negative,degradable mucus bacterium of the Verrucomicrobiota phylum and Akkermansiaceae family, with only one species identified,Akkermansia muciniphila.Akkermansia muciniphilawas associated with healthy mucosa and strengthening the integrity of the epithelial cell layer, which could contribute to intestinal health [11,40]. In our study, the relative abundances of Verrucomicrobiota phylum and Akkermansiaceae family in the gut microbiota of the PEW group were higher than those of the DSS groups, which may be due to the higher relative abundance ofAkkermansiagenus in the PEW group.The relative abundance of Lachnospiraceae, Muribaculaceae,Blautiaand Ruminococcaceae was reduced and that of the harmful bacteria Proteobacteriaincreased in the DSS group when compared to the control group, which was consistent with previous studies [14,37,51].Proteobacteria are considered pathogenic bacteria with the production of endotoxins [52]. The increase of endotoxin content in the intestine can lead to various conditions such as intestinal inflammation,immune disorders and intestinal mucosal dysfunction [53].Also Yu et al. [13] found that the consumption of PEW by rats significantly reduced the relative abundance of Proteobacteria when compared to ceca from rats fed regular duck egg white.
It was reported thatBlautiaplayed an important role in preventing inflammation by upregulating intestinal regulatory T cells and producing SCFAs [54].Lachnospiraceae is considered a beneficial microorganism for its ability to degrade toxins and produce propionic acid [55]. Ruminococcaceae, a bacterium producing butyrate may improve colitis [18,29,38], by decreasing intestinal cell damage and secretion of pro-inflammatory cytokines in the colon. Muribaculaceae is the main gut microbiota identified in healthy individuals, producing acetic acid and propionic acid through fermentation [56]. SCFAs(including acetic acid, propionic acid and butanoic acid), produced by the intestinal floras, could alleviate inflammation by inhibiting the NF-κB pathway and then inhibiting the release of inflammatory cytokines such as IL-12, TNF-α and IL-6 [57]. Butanoic acid, in particular, plays an important role in human health, providing directly energy to the intestinal epithelium, promoting the intestinal immune response, improving the immune function in the intestine, and thus overall protecting the intestine [58]. In our study, the concentrations of acetic acid, butanoic acid and total short-chain fatty acid levels in the ceca of mice suffering from colitis were much lower than those in mice of the control group. Conversely, the PEW treatment increased the concentrations in acetic acid, propionic acid,butanoic acid and total short-chain fatty acid levels in the ceca.Simultaneously, compared with the DSS group, the treatment with PEW increased the relative abundances of Lachnospiraceae,Muribaculaceae,Blautiaand Ruminococcaceae, and decreased the relative abundance of Proteobacteria (harmful bacteria). Overall,our results indicated that PEW treatment alleviated the signs of colitis in mice by regulating the composition of the gut microbiota and its metabolites.
Some polypeptides and amino acids derived from avian eggs have been proven to exhibit anti-oxidant and anti-colitis activities [28,59].As found in previous studies [26,60,61], the pretreatment of Caco-2 cells with egg yolk-derived phosphopeptides and simulated gastrointestinal digest from preserved egg whites could inhibit oxidative stress and achieve anti-inflammatory effects. After gastrointestinal digestion, preserved egg white proteins were mostly decomposed into polypeptides and free amino acids, which were more likely to interact with target biomolecules such as membrane receptors in the intestine compared with other macromolecular active components [21,62]. The peptides DEDTQAMPFR (DR-10),SLSFASR (SR-7), MLGATSL (ML-7) and MSYSAGF (MF-7)derived from SGD-PEW were shown to alleviate the degree of colitis in mice [28]. In addition, peptides with the amino acids Trp, Tyr, His,and Met, had stronger anti-oxidant activitiesin vitroandin vivo[63].Therefore, the anti-inflammatory and anti-oxidant properties of polypeptides and amino acids derived from egg whites may contribute to ameliorating colitis symptoms. On the other hand, preserved eggs contained some undigested high-molecular-weight polypeptides afterin vitrosimulated digestion [64]. The undigested proteins and polypeptides could be further utilized by gut microbiota in the large intestine to supply basic substances (such as amino acids and nitrogen) for the growth and metabolism of bacteria, as additional substrates for SCFAs production [65], which explains the increase in fecal SCFAs levels in the PEW group.
5. Conclusions
Our study demonstrated that the possible mechanisms of PEW treatment on improving DSS-induced colitis were mainly through the regulation of the gut microbiota and its metabolites, the inhibition of pro-inflammatory cytokines, the regulation of oxidative stress, and the inhibition of NF-κB and MAPK activation. PEW did not only reduce the inflammatory cells infiltration in the colon tissues of mice, but also regulated the oxidative stress to repair the oxidative damage in the colon tissues. PEW also inhibited the secretion and gene expression of a large number of pro-inflammatory cytokines (includingTNF-α,IL-6andIL-1β) by suppressing the phosphorylation of NF-κB p65 and p38 MAPK proteins, thereby reducing inflammation. Finally, PEW also alleviated DSS-induced colitis by improving the composition of intestinal microbiota and enhancing the production of SCFAs in the cecum of mice. In the future, further studies determining the role of inflammation-related signaling pathways as well gut microbiota and their metabolites in the anti-inflammatory effects of specific active ingredients of PEW are needed.
Conflicts of interest
The authors declare that there are no conflicts of interest.
Acknowledgements
This work was financially supported by the Chinese National Natural Science Funds (31772043), and the Fundamental Research Funds for the Central Universities (2662018JC021).
Appendix A. Supplementary data
Supplementary data associated with this article can be found, in the online version, at http://doi.org/10.1016/j.fshw.2022.07.021.
- 食品科学与人类健康(英文)的其它文章
- Wine, beer and Chinese Baijiu in relation to cardiovascular health:the impact of moderate drinking
- Comparative analysis of physicochemical properties, ginsenosides content and α-amylase inhibitory effects in white ginseng and red ginsen
- Monitoring and identif ication of spoilage-related microorganisms in braised chicken with modif ied atmosphere packaging during refrigerated storage
- Effect of cooking processes on tilapia aroma and potential umami perception
- Formation mechanisms of ethyl acetate and organic acids in Kluyveromyces marxianus L1-1 in Chinese acid rice soup
- Volatile prof ile and multivariant analysis of Sanhuang chicken breast in combination with Chinese 5-spice blend and garam masala

